Bihar Board Class 12 Physics Solutions Chapter 13 Nuclei Textbook Questions and Answers, Additional Important Questions, Notes.
BSEB Bihar Board Class 12 Physics Solutions Chapter 13 Nuclei
Bihar Board Class 12 Physics Nuclei Textbook Questions and Answers
You may find the following data useful in solving the exercises:
e = 1.6x 10-19C,
\(\frac{1}{\left(4 \pi \varepsilon_{0}\right)}\) = 9 x 109Nm2/C2,
I MeV = 1.6 x 10-13J,
1 year = 3.154 x 107s
mH = 1.007825 u,
m (42He) = 4.002603 u,
N = 6.023 x 1023 per mole
k = 1.381 x 10-23 J0K-1
lu = 931.5 MeV/c2
mn = 1.008665 u
me = 0.000548 u
Question 1.
(a) Two stable isotopes of lithium 63Li and 73Li have respective abundances of 7.5% and 92.5%. These isotopes have masses 6.01512u and 7,01600u, respectively. Find the atomic weight of lithium.
(b) Boron has two stable isotopes, 105B and 115B Their respective masses are 10.01294 u and 11.00931 u, and the atomic mass of boron is 10.811 u. Find the abundances of 105B and 115B.
Answer:

Let A be the atomic weight of Lithium.
∴ A is given by the formula,

(b) Here. A = Atomic weight of B = 10.811 u.
Let % and (100 – a)%betheabundancesof 105B and 115B isotopes respectively

∴ A give by
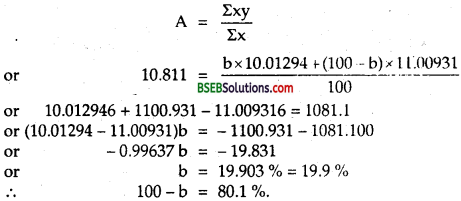
Question 2.
The three stable isotopes of neon : 2010Ne 2110Ne and 2210Ne have respective abundances of 90.51 %, 0.27 % and 9.22 %. The atomic masses of three isotopes are 19.99 u, 20.99 u and 21.99 u, respectively. Obtain the average atomic mass of neon.
Answer:

Let A = average atomic mass of neon = ?
A is given by formula, m = \(\frac{\Sigma x y}{\Sigma x}\)

Question 3.
Obtain the binding energy in MeV of a nitrogen nucleus (147N), given m (147N) = 14.00307 u.
Answer:
147N is made up of 7 protons and 7 neutrons.
Here,
mH = 1.00783 u
mn = 1.00867 u
mN = 14.00307 u
B.E. = ?
∴ Mass of nucleons forming nucleus.
= 7mH + 7mn
= 7 x 1.00783 u + 7 x 1.0086 7 u
= 7.05481 + 7.06069 = 14.11550 u.
If ∆m be the mass defect, then
∆m = mass of nucleons – mass of nucleus
= 14.11550 u -14.00307 u
= 0.11243 u
![]()
Using Formula,
B.E. = ∆m x 931 MeV
= 0.11243 x 931 MeV = 104.67 MeV = 104.7 MeV.
Question 4.
Obtain the binding energy of the nuclei 5626Fe and 20983Bi in units of MeV from the following :
m (5626Fe) = 55.934939 u m (20983Bi) = 208.980388 u
Answer:
Here, mass of hydrogen atom, mH 1.007825 a.m.u.
mass of neutron, mn = 1.008665 a.m.u.
(i) 5626Fe nucleus contain 26 protons and (56 – 26) = 30 neutrons
∴ Mass of 26 protons = 26 x 1.007825 am.u = 26.20345 a.m.u.
Mass of 30 neutrons = 30 x 1.008665 am.u = 30-25995 a.m.u.
Total mass of 56 nucleons = (26.20345 + 30.25995) a.m.u
= 56.46340 a.m.u.
Mass of 5626Fe nucleus = 55.934939 a.m.u.
∆m = mass defect is given by
∆m = mass of nucleons – mass of nucleus of 5626Fe.
= (56.46340 – 55.934939) a.m.u
= 0.528461 a.m.u.
Total binding energy = 0.528461 a.m.u
= 0.528461 x 931.5 MeV = 492.26 MeV.
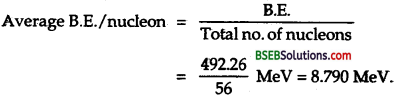
(ii) 20983Bi nucleus contains 83 protons and (209 – 83) = 126 neutrons.
∴ Total mass of nucleons = 83 x mH +126 mn
= 83 x 1.007825 + 126 x 1.008665 = 83.649475 +127.091790 a.m.u. = 210.741260 a.m.u.
Mass of 20983B nucleus = 208.980388 a.m.u.
∴ Mass defect is given by
∆m = mass of nucleons – mass of nucleus of 20983Bi
= (210.741260 – 208.980388) a.m.u. or ∆m = 1.760872 a.m.u.
∴ B.E. of 20983Bi = 1.760872 x 931.5 MeV = 164026 MeV
Average B.E/nucleon = \(\frac{1640.26}{209}\) – MeV
= 7.84MeV/nucleon
Question 5.
A given coin has a mass of 3.0 g. Calculate the nuclear energy that would be required to separate all the neutrons and protons from each other. For simplicity assume that the coin is entirely made of 6329Cu atoms (of mass 62.92960 u).
Answer:
Mass of Copper coin = 3g
Symbol of Copper nucleus ZXA = 29Cu63
A copper nucleus has 29 protons each of mass 1.00783 u and it has 63 – 29 = 34 neutrons each of mass 1.00867 u
In 63 g of copper, No. of copper atoms = Avogadro Number
= N = 6.023 x 1023
∴ No. of atoms in 1 g = \(\frac{6.023 \times 10^{23}}{63}\)
and No. of atoms in 1 g = \(\frac{6.023 \times 10^{23}}{63} \times 3\)
For 1 atom of copper, mass of nucleons = Mass of 29 protons + Mass of 34 neutrons
= (29 x 1.00783 + 34 x 1.00862) amu
Mass of Copper nucleus = 62.9260 u
Mass defect, ∆m = 29 x 1.00783 + 34 x 1.00862 – 62.9260 amu (for 1 copper nucleus)
In 3 gram of copper, no. of copper nuclei = 2.868 x 1022
Mass defect for 3 gram of copper
= (29 x 1.00783 + 34 x 1.00862 – 62.9260) x 2.868 x 1022
= (83.82 + 98.36 -180.48) x 1022 amu
But 1 amu = 9.311 MeV
∴ Energy equivalent to mass defect = 1.70 x 1022 x 931
= 1582.7 x 1022 MeV
This much energy is required to break copper nuclei into constituent nucleons (protons and neutrons)
∴ Energy Required = 1582.7 x 1022 MeV.
= 1.583 x 1025 MeV = 1.583 x 1025 x 1.6 x 10-13J = 2.533 x 1012 J.
![]()
Question 6.
Write nuclear reaction equations for
(i) α – decay of 22688Ra
(ii) α – decayof 24294Pu
(iii) β – decay of 3215P
(iv) β-decay of 21083Bi
(v) β+ – decay of 116 C
(vi) β+ – decay of 9743 Tc
(vii) Electron capture of 12054Xe
Answer:
α – decay of Radium.
(i) α – decay of 22688Ra → 22286Rn + 42He (α-particle).
Parent nucelus decays into radn.
(ii) α – decay of Pu.
24294Pu → 23892U + 42He (α-particle).
(iii) β– – decay of P
3215P → 3216S + -1e° + v–
i.e.,β-decay is accompanied by relese of antineutrino.
(iv) β– – decay of 21083Bi
21083Bi → 21084P + –1e° + v–
(v) β+ – decay of 116C :
116C → 5C11 + +1e° + v
P+-decay of is accompanied by the release of neutrino.
(vi) β+ – decay of 9743Tc :
9743Tc → 9742Mo + +1e° + v
(vii) Nuclear reaction equation for electron capture of 12054Xe is given by
12054Xe + +1 e0 → 12053I + v
Question 7.
A radioactive isotope has a half-life of T years. How long will it take the activity to reduce to a) 3.125 %, b) 1 % of its original value?
Answer:
Let N0 = initial activity
N = activity after time
T = half-life of the radioactive isotope
λ = disintegration constant
∴ initial activity A0 is given by
A0 = λ N0
Also we know that
N = N0 (\(\left(\frac{1}{2}\right)^{\frac{t}{T}}\))
(a) Here A = 3.125 % of A0 = \(\frac{3.125}{100}\) A0
= \(\frac{3125}{100 \times 1000}\) A0
= \(\frac{1}{32}\) A0 ………….(1)
∴ Activity, A = – \(\frac{\mathrm{d} \mathrm{N}}{\mathrm{d} \mathrm{t}}\) = λ N = λN0(\(\left(\frac{1}{2}\right)^{\frac{t}{T}}\))
∴ A = A0 (\(\left(\frac{1}{2}\right)^{\frac{t}{T}}\)) ……….(2)
∴ from (1) and (2) we get,
\(\frac{1}{32}\) = (\(\left(\frac{1}{2}\right)^{\frac{t}{T}}\))
or \(\frac{1}{32}\)5 = (\(\left(\frac{1}{2}\right)^{\frac{t}{T}}\))
or 5 = \(\frac{t}{T}\)
or t = 5 T years.
After time 5 half periods activity reduces to 3.125 % of initial activity.
![]()
(b) Here, A = 1 % of A0 = \(\frac{1}{100}\)A0
or \(\frac{A}{A_{0}}=\frac{1}{100}\) ………..(1)
Also we know that \(\frac{\mathrm{A}}{\mathrm{A}_{0}}\) = e-λt …………(2)
∴ From (1) and (2), we get
\(\frac{1}{100}\) = e-λt or eλt = 102. 102
Taking log on both sides, we get
λt = log e102 = 2 x 2.303 log1010 = 4.606
∴ t = \(\frac{4.606}{\lambda}\) ………..(3)
Also we know that, λ = \(\frac{0.693}{\mathrm{~T}}\) …….(4)
∴ From (3) and (4),,we get
t = \(\frac{4.606}{\left(\frac{0.693}{\mathrm{~T}}\right)}=\frac{4.606}{0.693}\) T
= 6.65 T years.
Question 8.
Thenormalactivity of living carbon – containing matter is found to be abouil5 decays percmmrte for every gram of carbon. This activity arises from the small proportion of radioactive 146C present with the stable carbon isotope 146C. When the organism is dead, its interaction with the atmosphere (which maintains -the above equilibrium activity) ceases and its activity begins to drop. From the known half-life (5,730 years) of 146C, and the measured activity, the age of the specimen can be estimated. This is the principle of 146C dating used in archaeology. Suppose a specimen from Mohenjodaro gives an activity of 9 decays per minute per gram of carbon. Estimate the approximate age of the Indus Valley Civilisation.
Answer:
`Here, let the no. of C14 atoms per gram were N0 at t = 0 (initially), when its activity was 15 decays perminute per gram. Today after time t the no. of C14 atom per gram left is N and it shows an activity of a decays per minute per gram.
i.e., normal activity, R0 = 15 decays / min.
Present activity, R – 9decays/min.
\(\mathrm{T}_{\frac{1}{2}}\) = 5,730 years
Aget = ?
Also we know that activity is directly proportional to the number of radioactive atoms, so \(\frac{N}{N_{0}}=\frac{R}{R_{0}}=\frac{9}{15}\)
Also we knoe that
\(\frac{N}{N_{0}}\) = e-λt
or \(\frac{9}{15}\)= e-λt
or e-λt = \(\frac{5}{3}\) = 1.6667
λt logee = loge 1.6667
λt = 2.303 log10 1.6667
t = \(\frac{0.5109}{\lambda}\) …………(1)
Also we know that decay constant is given by
λ = \(\frac{0.693}{\mathrm{~T}_{\frac{1}{2}}}=\frac{0.693}{5,730}\) ………..(2)
∴ From (1) and (2), we get
t = \(\frac{0.5109}{0.693}\) x 5,730 = 4225.15 years.
Question 9.
Obtain the amount of 6027Co necessary to provide a radioactive source of 8.0 mCi strength. The half-life of 146Co is 5.3 years.
Answer:
Here, R = activity = strength of radioactive source = 8.0 m Ci = 8 x 10-3 x 3.7 x. 1010 disintegrations/sec.
= 2.96 x 108 disintegrations/sec (∵ 1 Ci = 3.7 x 1010 disintegrations s-1).
\(\mathrm{T}_{\frac{1}{2}}\) = half-life of 6027Co = 5.3 years
s = 5.3 x 365 x 24 x 60 x 60 s = 1.67 x 108 s.
Nt = no of nucli present at time = ?
Using relation,
R = – \(\frac{\mathrm{d} \mathrm{N}}{\mathrm{dt}}\) = – \(\frac{\mathrm{d}}{\mathrm{dt}}\)(N0e-λt)
= + λ N0e-λt
![]()
Also know that
λ = \(\frac{0.693}{\mathrm{~T}_{1}}=\frac{0.693}{1.67 \times 10^{8}}\) s
∴ From (1) and (2), we get
\(\frac{0.693}{1.67 \times 10^{8}}\) Nt = 2.96 x 108 1.67 x10s 1
Nt = \(\frac{2.96 \times 10^{8} \times 1.67 \times 10^{8}}{0.693}\)
= 7.133 x 1016
A mount of 146Co = ?
We know that 60 g of Cobalt contains 6.023 x 1023 cobalt atoms or nuclei.
i e., 6.023 x 1023 atoms of Co have mass = 60 g
1 atom of Co have mass = \(\frac{60}{6.023 \times 10^{23}}\) g
∴ 7.133 x 1016 atoms of Co have mass = \(\frac{60}{6.023 \times 10^{23}}\) x 7.133 x 1016
i.e.,Amount of 6027C0 needed = 7.11 μg.
Question 10.
The half-life of 9038Sr is 28 years. Whal is the ‘ disintegration rate of 15 mg of this isotope?
Answer:
Here, \(\mathrm{T}_{1 / 2}\) = Half-life of 9038Sr = 28.0 years = 28 x 365 x 24 x 3,600 s = 88.3 x 107 s.
Amount of 9038Sr = 15 mg = 15 x 10-3g.
Now 90 gm of Sr has atoms = 6.023 x 1023.
∴ 1 g of Sr will have atom = 6.023 x 1023
∴ Number of atoms of 30Sr in 15 mg is given by
= \(\frac{6.023 \times 10^{23}}{90}\) x 15 x 10-3
= 1.004 x 1020
∴ Nt = 1.004 x 1020
Also using λ = \(\frac{0.693}{\mathrm{~T}_{\frac{1}{2}}}\) we get
λ = \(\frac{0.693}{88.3 \times 10^{7}}\) s-1
R = ?
Using the relation, R = XN1, we get
R = \(\frac{0.693}{88.3 \times 10^{7}}\) x 1.004 x 1020
= 7.879 x 10-3 x 1013
= 7.879 x 1010 disinteg rafions s-1
= 7.879 x 1010Bq.
= \(\frac{7.879 \times 10^{10}}{3.7 \times 10^{10}}\) Ci (∵ 1 Ci = 3.7 x 1010 Bq)
= 2.13 Ci
∴ Disintegration rate = 2.13 Ci or 7.879 x 1010Bq.
Question 11.
Obtain approximately the ratio of the nuclear radii of the gold isotope 19779Au and the silver isotope 19747Ag .
Answer:
We know that for a spherical nucleus, its radius is given by
R = R0 A\(A^{\frac{1}{3}}\) ………(1)
Where A is the mass number of the nucleus, RQ is an empirical constant.
Let R1, A1 be the nuclear radius and mass number of the 19779Au isotope.
Also let R2, A2 be the nuclear radius and mass number of 19747Ag isotope.
∴ A1 = 197, A2 = 107
∴ Using (1)
R1 = A0\((197)^{\frac{1}{3}}\) ………(2)
And R2 = A0\((197)^{\frac{1}{3}}\) ……….(3)
Dividing (2) by (3), we get
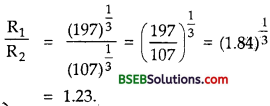
Question 12.
Find the Q-value and the kinetic energy of the ifted α-particle in the α-decay of (a) \(\frac{226}{88} \mathrm{Ra}\) (b) \(\frac{220}{86} \mathrm{Rn}\)
Given m (\(\frac{226}{88} \mathrm{Ra}\)) 226.02540 u,m (\(\frac{226}{88} \mathrm{Rn}\)) = 222.01750 u
m (\(\frac{222}{88} \mathrm{Rn}\)) = 220.01137 u, m (\(\frac{216}{84} \mathrm{po}\)) 216.00189 u.
Answer:
(a) The process of a-decay of \(\frac{226}{88} \mathrm{Ra}\) can be expressed as :
\(\frac{226}{88} \mathrm{Ra}\) -» \(\frac{222}{86} \mathrm{Ra}\)Rn + 24He +Q ……….(1)
Q-value of the reaction is given by
![]()
When mN represents the nuclear masses of the respective nuclei.
In forms of atomic masses, eqn. (2) can be expressed as :
i
The K.E. of the emitted α-particle is given by
(K-E-)α = \(\frac{A-4}{A}\) Q
Here, A = 226, ∴ A – 4 = 226 – 4 = 222, = 4.9342 MeV
∴ (K.E.) α = \(\frac{222}{226}\) x 4.9342 MeV
= 4.8469 MeV
= 4.85 MeV.
(b) The process of α-decay of \({ }_{86}^{220} \mathrm{Rn}\) can be expressed as :
\({ }_{86}^{220} \mathrm{Rn}\) → \({ }_{84}^{216} \mathrm{Po}\) +αHe + Q ………..(1)
The Q-value of the reaction is given by :

Let Eα be the K.E. of the a-particle emitted.
∴Eα is given by
Eα = \(\frac{\mathrm{A}-4}{\mathrm{~A}}\) Q
Where A = 220, A – 4 = 216, Q = 6.41 MeV.
∴Eα is given by :
E = \(\frac{216}{220}\) = 6.41 MeV.
= 6.289
= 6.29 MeV.
![]()
Question 13.
The radionuclide 11C decays according to
116C → 115B + e+ + v : \(\mathbf{T}_{1 / 2}\) = 20.3 min
The maximum energy of the emitted positron is 0.960 MeV.
Given the mass values :
m (116C) = 11.011434 u and m (116B) = 11.009305 u,
calculate Q and compare it with the maximum energy of the positron emitted.
Answer:
The equation for the decay process is –
116C → 115B + \(+1 \mathrm{e}^{0}\) + v + Q
Where Q – value of the reaction or K.E. of α-particle. = mass defect in a.m.u.
= (Mass of 116C nucleus – Mass of 115B nucleus – mass of positron in a.m.u
= \(\left\lfloor\mathrm{m}_{\mathrm{N}}\left({ }_{6}^{11} \mathrm{C}\right)-\mathrm{m}_{\mathrm{N}}\left({ }_{5}^{11} \mathrm{~B}\right)-\mathrm{m}_{\mathrm{e}}\right\rfloor\) ………..(1)
Where mN stands for the nuclear mass of the element or particle.
∴ As 116C atom is made up of 116C nucleus and 6 electrons.
∴ Mass of 116C nucleus = mass of 116C atom – mass of 6 electrons
= 11 011434 a.m.u – 6 me ………..(2)
Similarly mass of 115B nucleus = mass of 115B atom – mass of 5 electrons.
– 11.009305 a.m.u – 5 me …….(3)
∴ From (1), (2) and (3), we get
Q = (11.011434 a.m.u. – 6me) – (11.009305 a.m.u. – 5me) -me]
= (11.011434 -11.009305) a.m.u. – 2me
= (11.011434 -11.009305) a.m.u. – 2 x 0.0005048 a.m.u.
= 0.001033 a.m.u.
= 0.001033 x 931.5 MeV = 0.962 MeV ……….(4)
Maximum kinetic energy of emitted position (β+) is 0.960 MeV ……..(5)
∴ From (4) and (5), we conclude that the Q-value of the reaction is comparable to the actual energy released in the decay process.
Q = ED + Ee + EV
Tire daughter nucleus is too heavy compared to e+ and v, so it carries neglible energy (Ed ≈ 0). If the kinetic energy (Ev) carried by the neutrino is minimum (i.e., zero), the psotron carries maximum energy, and this is practically all energy Q; hence maximum Ee ≈ Q).
![]()
Question 14.
The nucleus 2310Ne decays by β– emission. Write down the β-decay equation and determine the maximum kinetic energy of the electrons emitted. Given that :
m (2310Ne) = 22 994466 u
m (2310Na) = 22.089770 u.
Answer:
Here,mass of 2310Ne atom = 22,094466 u.
mass of 2311Na atom = 22 989770 u.
The β-decay equation of 2310 Ne is
2310 Ne → 2311 Ne + -1e° + v + EB
Where EB is the maximum kinetic energy of the emitted p-particle and is equal to the Q-value of the reaction and is given by
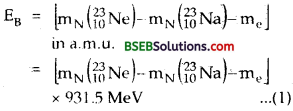
where mN (2310Ne) and mN (2311Na) are the nuclear masses of 2310Ne and 2311Na respectively. If m (2310Ne) and m (2311Na)be the atomic masses of 2310Ne and 2311Na respectively, then
mN (2310Ne) = m (2311Ne) – 10 me (∴ atom of Ne contains 10 electrons)
and mN (2311Na) = m (2311Na) – 11 me ( ∴atom of Na contains 11 electrons)
∴ From (1), Eβ = [ m(2311Ne) – 1oMe] – (2311Na – 11me] – me] in a.m.u
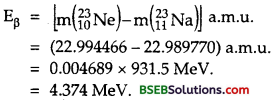
The energy released is shared by 2311Na nucleus and the electron -neutrino pair released. As electron-neutron pair is much lighter than 2311Na nucleus, practically whole of the energy released is carried by electron-neutrino pair. When neutrino gets zero energy, the electron will carry the maximum energy. So the maximum K.E. of the electron emitted is 4.374 MeV.
Question 15.
The Q value of a nuclear reaction A + b → C + d is defined by Q = [mA + mb + mC + md]C2 where the masses refer to the respective nuclei. Determine from the given data the Q-value of the following reactions and state whether the reactions are exothermic or endothermic.
(i) 11H + 31H → 21H + 21H
(ii) 126C + 126C → 2010Ne + 42He
Atomic masses are given to be
m (21H) = 2.014102 u
m (31H) = 3.016049 u
m (126C) = 12.000000 u
m (2010 Ne) = 19.992439 u
Answer:
(i) The given reaction is :
11H + 31H → 21H + 21H
∴ Q-value of the reaction is :
![]()
Where mN refers to nuclear masses.
m (11H) = mN (11H) + me or mN (11H) = m(11H) – me.
Similarly, mN (31H) = m(31H) – me
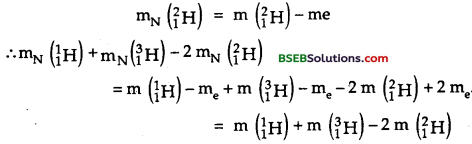
= 1.007825 + 3.016049 – 2 x 2.014102
= – 0.004339 a. m.u.
= -0.00433 x 1.66 x 10-27 kg.
∴ From (1), Q = – 0.00433 x 1.66 x 10-27 x (3 x 108)2
= – 6.46 x 10-13 J
= \(\frac{-6.46 \times 10^{-13}}{1.6 \times 10^{-13}}\) MeV = – 4.03 eV
Since the Q-value is negative, so the reaction is endothermic,
(ii) The given reaction is :
![]()
Q-value of the reaction is given by
![]()
Where mN refers to masses of respective nuclei and

∴ From (2)
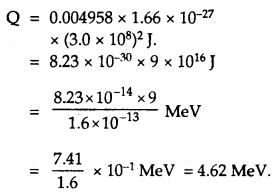
Since the Q-value is positive, so the reaction is exothermic.
Question 16.
Suppose, we think of fission of a 5626Fe nucleus into two equal fragments, 2813 Al . Is the fission energetically possible? Argue by working out Q of the process. Given m (5626Fe) = 55.93494 u and m (2813 Al) = 27.98191 u.
Answer:
The given reaction for the decay process is :
![]()
where Q-value of the reaction is given by.
Q = [mn + m(5626Fe) – 2m(2813Al)] x 931.5 MeV
= [1.00867 + 55.93494 – 2 x 27.98191] x 931.5 MeV
= (56.94361 – 55.96382) x 931.5
= 0.97979 x 931.5 MeV
= 912.67 MeV.
Although the Q-value is positive but the mass number (i.e., A) is not conserved in this process. Therefore the process of fission of 5626Fe cannot be possible.
Question 17.
The fission properties of 23994Pu are very similar to those of 23592U. The average energy released per fission is 180 MeV. How much energy, in MeV, is released if all the atoms in 1 kg of pure 23994Pu undergo fission?
Answer:
Here, average energy released per fission of 23994pu = 180 MeV.
Total energy released (MeV) = ?
Amount of 23994pu = 1 kg = 1,000 g
We know that 239 g of 23994pu contains = 6.023 x 1023 fissionable nuclei.
or 1g of 23994pu contains = \(\frac{6.023 \times 10^{23}}{239}\)
∴ No. of fissionable nuclei in 1,000 g of 23994pu
\(\frac{6.023 \times 10^{23}}{239}\) x 1,000
= 25.2 x 1023 nuclei.
∴ Total energy released in fission of 1 kg of Pu
= 180 x 25.2 x 1023 MeV
= 4536 x 1023 MeV
= 4.536 x 1026 MeV.
![]()
Question 18.
A1000 MW fission reactor consumes half of its fuel in 5.0 y. How much 235 JJ did it contain initially? Assume that the reactor operates 80 % of the time, that all the energy generated arises from the fission of 23592U and that this nuclide is consumed only by the fission process.
Answer:
Here, P = power of reactor = 1,000 MW = 103 x 106 W = 109 Js-1
t = time = 5 years = 5 x 365 x 24 x 3,600 s = 1.577 x 108 s.
If E be the amount of energy delievered by the reactor when it operates with 80 % of time, then
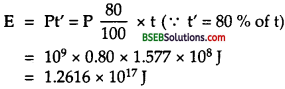
Q = energy generated per fission of23592U = 200 MeV.
= 200 x 106 x 1.6 x 10-19 J
= 3.2 x 1011 J.
Let n be the number of fissions occurred in 5 y when the reactor operates 80 % of the time.

Now 6.0 x 1023 atoms or fissions are produced by 235 g of 23592U
∴ Mass of 23592U consumed in five years is given by
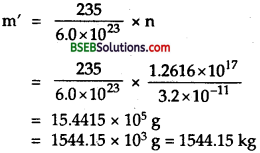
or m’ = half of the fuel consumed in 5 g.
∴ If M be the initial mass of 23592U, then
m = 2m’
= 2×1544.15
= 3088.3 kg.
Question 19.
How long can an electric lamp of 100 W be kept glowing by fusion of 2.0 kg of deuterium? Take the fusion reaction as ?
21H + 21H → 32He + n + 3.27 MeV
Answer:
Here, P – Power of lamp = 100 W.
m = mass of deuterium = 2.0 kg.
The fusion reaction is given by
21H + 21H → 32He + n + 3.27 MeV ……..(1)
From eq. (1), it is clear that 2 nuclei of 21H (deuterium) combine to give 3.27 MeV of energy.
Now 2 g of 21H contains 6.023 x 1023 nuclei.
∴ Numer of 21H nuclei in 2 kg of deterium = \(\frac{6.023 \times 10^{23}}{2 \times 10^{-3}}\) x 2
= 6.023 x 1026 nuclei.
Energy released in fusion of two nuclei of 21H = 3.27 MeV.
∴ Energy released in fusion of 6.023 x 1026 nuclei of it is given by
E = \(\frac{3.27 \times 6.023}{2} \times 10^{26}\) MeV
or E = \(\frac{3.27 \times 6.023}{2} \times 10^{26}\) x 1026 x 1.6 x 10-13 J = 15.76 x 1013J ………(2)
Let t = time for which the electric lamp can glow due to this energy = ?
∴ energy consumed by the lamp, E’ = Pt = 100 t ……….(3)
∴ According to law of conservation of energy
E’ = E
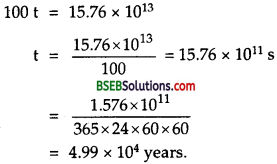
Question 20.
Calculate the height of the potential barrier for a head on collision of two deuterons. (Hint : The height of the potential barrier is given by the Coulomb repulsion between the two deuterons when they just touch each other. Assume that they can be taken as hard spheres of radius 2.0 fm.)
Answer:
Let r be the distance of closest approach of the two deutrons for head-on-collision.
Here r = 2 fm = 2 x 10-15 m.
e = charge on each deutron
= 1.6 x 10-19 C.
![]()
Let E be the initial mechanical energy of the two deutrons before collision.
∴ E = 2 K.E. …………(1)
where K.E. = Kinetic energy of each deutron. When the two deutrons stop, their energy is totally electric potential energy (= U) given by
U = Fe. r
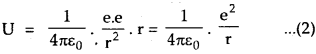
∴ according to the law of conservation of energy,
E = U

∴ height of potential barrier = K.E. = 360 KeV.
Question 21.
From the relation R = R0 \(\mathbf{A}^{\mathbf{1 / 3}}\), where R0 is a constant and A is the mass number of a nucleus, show that the nuclear matter density is nearly constant (t.e., independent of A).
Answer:
The expression of the radius of nucleus is given by
R = R0 \(\mathbf{A}^{\mathbf{1 / 3}}\) ………..(1)
Where R0 is a constant and A is the mass number of a nucleus. This can be used to calculate the density of the nucleus, mass of the nucleus of the atom = A a.m.u.
= A x 1.66 x 10-27 kg.
If V be the volume of the nucleus, then

Using R0 = 1 .1 x 10-15m, eqn. (2) becomes
V = \(\frac {4}{3}\)π x (1.1 x 10-15)3 A ……….(3)
If δ be the density of the nucleus, then

Thus from equation (4) we see that ρ is independent of A, hence we conclude that p is nearly constant for all nuclei.
Question 22.
For the β+ (positron) emission from a nucleus, there is another competing process known as electron capture (electron from an inner orbit, say, the K-shell, is captured by the nucleus and a neutrino is emitted).
![]()
Show that if β+ emission is energetically allowed, electron capture is necessarily allowed but not vice-versa.
Answer:
The β+ emission from a nucleus AZXcan be represented as :
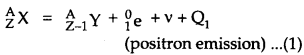
The other competing process of electron capture may be represented as :
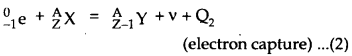
The energy released Qj in Eq. (1) is given by
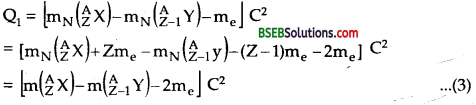
Here mN denotes the mass of nucleus and m denotes the mass of the atom.
Similarly from Eq. (2),
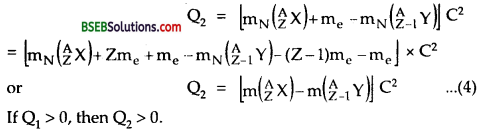
i.e., If positron emission is energetically allowed, electron capture is necessarily allowed. But Q2 > 0 does not necessarily mean Q1> 0. Hence the reverse is not true.
Bihar Board Class 12 Physics Nuclei Additional Important Questions and Answers
Question 23.
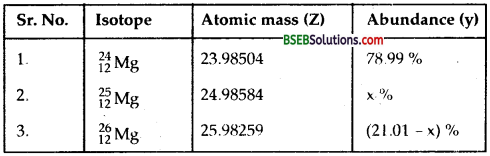
In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their masses are 2412Mg (23.98504 u), 2512Mg (24.98584 u) and 2612Mg (25.98259 u). The natural abundance of 2412Mg is 78.99 % by mass. Calculate the abundances of other two isotopes.
Answer:
Here, average atomic mass of magnesium = 24.312 u.
Let x % be the natural abundance of 2512Mg
Natural abundance of 2412Mg = 78.99 %
∴ Natural abundance of 2412MgMg = (100 – x – 78.99) % = 21.01 % – x %
We know that average atomic mass is given by the relation

i.e Natural abundance of 2412Mg = 9.30 %
∴ natural abundance of 2612Mg = (100 – 9.30 – 78.99) %
= 11.71%
Question 24.
The neutron separation energy is defined as the energy required to remove a neutron from the nucleus. Obtain the neutron separation energies of the nuclei 4120Ca and 2713Al from the following data :
m (4020Ca) = 39 962591 u
m (4120Ca) = 40.962278 u
m (2613Al) = 25.986895 u
m (2713Al) = 26.981541 u
mn = 1.008665 u.
Answer:
(i) Neutron separation energy of 4120Ca – When a neutron is separated from 4120Ca -we are left with 4020Ca Thus the nuclear reaction is given by
4120Ca → 4020Ca + 10n
∴ Mass defect is given by
∆m – m (4020Ca) +mn – m (4120Ca)
= [39.962591 + 1.008665 – 40.962278]
= 0.008978 a.m.u.
Thus neutron separation energy Sn is given by
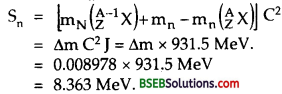
(ii) Neutron separation of 2713Al – When a neutron is separated from 2713Al, we are left with 2713Al, Thus the nuclear reaction is given by :
2713Al + 2613Al + 10n
∴ Mass defect is given by :
∆m = m (2613A1) + mn – m (2713Al)
= 25.986895 + 1.008665 – 26.981541
= 26.99556 – 26.981541
= 0.014019 a.m.u.
Thus the neutron separation energy is given by Sn = Am a.m.u.
Sn = ∆m a.m.u.
= ∆m x 931.5 MeV
= 0.014019 x 931.5 MeV
= 13.06 MeV.
![]()
Question 25.
A source contains two phosphorous radio nuclides 3215p (\(\mathrm{(T}_{\mathbf{1} / 2}\) = 14.3 d) and 3|p (\(\mathrm{(T}_{\mathbf{1} / 2}\) = 25.3 d). Initially, 10 % of the decays come from 3315p. How long one must wait until 90 % do so?
Answer:
Let R01 and R02 be the initial activities of 3315p and 3215 p respectively.
Also Let R1 and R2 be their respective activities at any instant t.,
∴ tota1 in’itial activity = R01 + R02 ……….(1)
Total activity of time t = R1 + R2 ………..(2)
Here, R01 = 10% of total activity

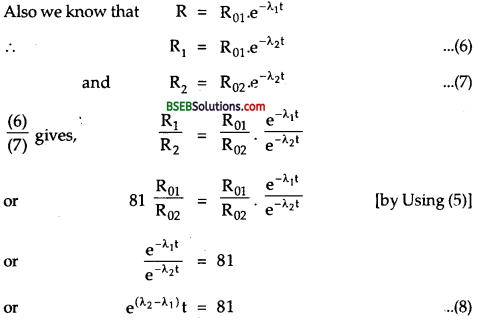
Taking loge on both sides of equation (8), we get
log e\(\left(\lambda_{2}-\lambda_{1}\right)\)t – loge 81.
or (λ2 – λ1)t = 2.303 log10 81 ……(9)
Also we know that
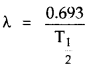
Let (\(\mathrm{T}_{1 / 2}\)), and (\(\mathrm{T}_{1 / 2}\))2 be he Hall-life time of 3315 p and 3315 prespectively.

From (9), (11) and (12), we get
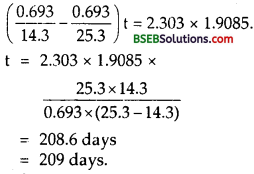
Question 26.
Under certain circumstances, a nucleus can decay by emitting a particle more massive than an a particle. Consider the following decay processes :
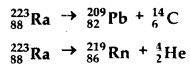
Calculate the Q-values for these decays and determine that both are energetically allowed.
Answer:
The given decay process for 22315Ra for 146C emission is
22315Ra → 20982Pb + 146C + Q ……….(1)
where Q-value of the reaction is given by
![]()
where mN stands for nuclear mass.
∴ Q in terms of atomic masses ‘m’ is given by

The decay process for 22315Ra for 42He emission is given by
22315Ra → 21986Rn + 42RaHe + Q ………(2)
Where Q-value of the reaction is given by

Question 27.
Consider the fission of 23892U fast neutrons. In one fission event, no neutrons are emitted and the final end products, after the beta decay of the primary fragments, are 14058Ce and 9944Ru . Calculate Q for this fission process. The relevant atomic and particle masses are .
m(23892U) = 238.05079 u
m (14058Ce) = 139.90543 u
m(9944Ru) = 98.90594 u
Answer:
The fission reaction may be written as :
23892U + 10n → 14058Ce + 9944Ru + Q
where Q-value of the reaction is given by
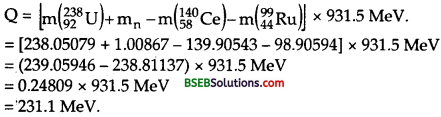
Question 28.
Consider the D – T reaction (deuterium-tritium fusion)
21H + 31H → 42H + n
(a) Calculate the energy released in MeV in this reaction from the data :
m (21H) = 2.014102 u
m (31H) = 3.016049 u
(b) Consider the radius of both deuterium and tritium to be approximately 2.0 fm. What is the kinetic energy needed to overcome the coulomb repulsion between the two nuclei? To what temperature must the gas be heated to initiate the reaction?
(Hint: Kinetic energy required for one fusion event = average thermal kinetic energy available with the interacting particles = 2 (3kT/2); k = Boltzman’s constant, T = absolute temperature.)
Answer:
(a) Here, m (21H) = 2.014102 a.m.u
m (31H) = 3.016049 a.m.u.
m (42He) – 4.002603 a.m.u.
mn = 1.008665 a.m.u.
The D – T fusion reaction is given as :
21H + 31H -» 42He + 10n + Q
where Q-value of the reaction is given by,

= [2.014102 + 3.016049 – 4.002603 – 1.008665] x 931.5 MeV
= 0.018883 x 931.5 MeV = 17.59 MeV.
(b) Here, r = radius of 21H or 31H= 2 fm = 2 x 10-15 m
∴ 2r = distance between 21H or 31H when the two nuclei almost come in contact with each other
charge on each i.e., q1 = q2 = 1.6 x 10-19 C.
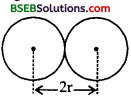
The P.E. of the D-T system, when the two nuclei almost come in contact with each other is given by :

The kinetic energy needed to over come Coulomb’s repulsion is equal to the P.E.
i.e. The required K.E. = 5.76 x 10-14J.
Also we know that kinetic energy required for one fusion event = average thermal kinetic energy available with the interacting particles
= 2 x \(\frac {3}{2}\) kT = 3 kT
where T = absolute temperature = ?, k = Boltzman constant = 1.38 x 1023 JK-1
or 5.74 x 10-14 = 3 kt
T = \(\frac{5.76 \times 10^{-14}}{3 \times 1.38 \times 10^{-23}}\) = 1.391 x 109
Question 29.
Obtain the maximum kinetic energy of β-particles, and the radiation frequencies of ydecays in the decay scheme shown in fig. You are given that m(198Au) = 197.968233 u
m(198Hg) = 197.966760 u
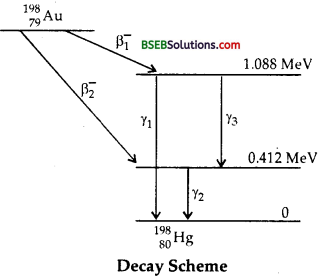
Fig. Decay Scheme
Answer:
Here, E1 = energy of ground state = 0
E2 = energy of Ist excited state = 0.412 MeV = 0.412. x 1.6 x 10-13 J
E3 = energy of 2nd excited state = 1.088 MeV = 1.088 x 1.6 x 10-13J.
The total energy released for the transformation of 19879Au to
19880Hg by β1 and β2 decay can be formed by considering the energies of γ-rays. .
Let v1, v2 and v3 be the frequencies of the γ-ray photons emitted i.e., y1, y2 and y3 respectively.

The emission of β1– decay may be expressed as :
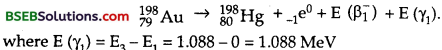
and the maximum kinetic energy is given by
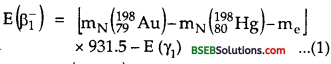
where mN (19879Au) and mN (19880Hg) are the masses of nuclie of Au and Hg respectively which can be expressed in terms of the masses of Au and Hg atoms. Thus eq. (1) can be written as :
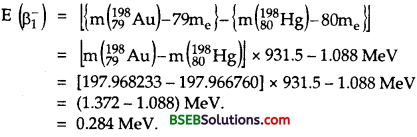
The emission of β2– decay may be expressed as :
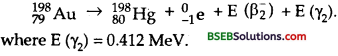
and the maximum kinetic energy E (β2–) is given by
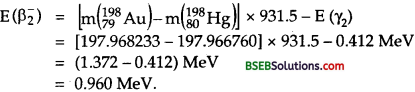
Question 30.
Calculate and compare the energy released by (a) fusion of 1.0 kg of hydrogen deep within Sun and (b) the fission of 1.0 kg of in a fission reactor.
Answer:
(a) The fusion reaction taking place in the sun is given by :
![]()
i.e., 4 hydrogen atoms combine to produce 26 MeV of energy per event.
Mass of hydrogen taken = 1 kg = 1,000 g.
Now 1 g of hydrogen contains 6.023 x 1023 nuclei.
∴ 1,000 g of hydrogen contains = 6.023 x 1023 x 103 nuclei.
= 6.023 x 1026 nuclei.
∴ Total energy released in fusion of all these nuclei
= \(\frac {26}{4}\) MeV x 6.023 x 1026
= 39.15 x 1026MeV.
(b) The nuclear fission reaction of 235 XJ is given by

In fission of single 23592U nucleus, the en ergy released = 200 MeV.
Mass of Uranium taken = 1kg = 1,000 g
We know that 235 g of 23592U has 6.023 x 1023 atoms or nuclei.
∴ Number of nuclei in 1 kg of 23592U
= \(\frac{6.023 \times 10^{23}}{235}\) x 1,000 atoms.
2.56 x 1024 atoms.
∴ Total energy released in fission of all 23592U nuclei is
1 kg = 200 x 2.56 x 1024 MeV.
= 5.12 x 1026 MeV.
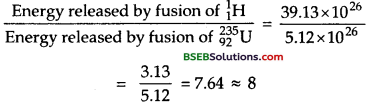
i.e. energy released per kg of fuel in fusion is about 8 times the energy released per kg of fuel in fission.
Question 31.
Suppose India had a target of producing by 2020 AD, 200,0 MW of electric power, ten percent of which was to be obtained from nuclear power plants. Suppose we are given that, on an average, the efficiency of utilization (i.e., conversion to electric energy) of thermal energy produced in a reactor was 25 %. How much amount of fissionable uranium would our country need per year by 2020? Take the heat energy per fission ot to be about 200 MeV.
Answer:
Here total targeted electric power = 200,00 MW = 2 x 105 MW.
= 2 x 105 x 106 W = 2 x 1011 W.
Total nuclear power target
= 10 % of 2 x 1011 W
= \(\frac {10}{100}\) x 2 x 1011 = 2 x 1010W.

∴ Total energy required per year by 2020 is given by
E = P x t
= 8 x 1010 x 1 year
= 8 x 1010 x 365 x 24 x 60 x 60 J
= 2.523 x 1018J.
Energy produced for fission of 235U= 200 MeV.
= 200 x 1.6 x 10-13J = 3.2 x 10-11 J.
∴ No. of fission (n) required per year for the generation of E is given by
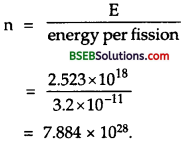
Now mass of 6.023 x 1023 atoms of 235U = 235 g = 235 x 10-3 kg
∴ Mass of 235 u required to produce 7.884 x 1028.
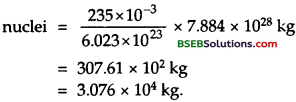
Very Short Answer Type Questions
Question 1.
Define binding energy of a nucleus.
Answer:
It is defined as the amount of work done to separate the nucleons an infinite distance apart from each other so that they no longer interact with each other.
Or
It is defined as the energy equivalent to the mass defect of the nucelus. Mathematically,
B.E. = ∆mc2 (J) = ∆m x 931.5 MeV.
Where ∆m – mass defect of nucleus, c = 3 x 108 ms-1 = speed of light.
Question 2.
Define the following:
(a) mass defect
(b) Binding energy per nucleon.
(c) Packing fraction of a nucleus.
Answer:
(a) Mass defect – It is defined as the difference between the sum of the masses of nucleons constituting a nucleus and the rest mass of the nucleus.
(b) B.E. per nucleon – It is defined as the average energy required to extract one nucleon from the nucleus.
![]()
(c) Packing fraction of a nucleus – It is defined as the mass defect per nucleon of the nucleus.
![]()
Question 3.
Define : (a) Isotones, (b) Isotopes, (c) Isobars.
Answer:
(a) Isotones – They are defined as the atoms whose nuclei have the same number of neutrons, e.g. 94Be and 105B, 2311Na and 2412Mg 136C and 147N.
(b) Isotopes – They are defined as the atoms of an element having same atomic number but the different mass number 168o and 178O.
(c) Isobars – They are defined as the atoms of the different elements having the same mass number but different atomic numbers.
e-g- 31H and 32He, 73Li and 74Be, 157N and 158Oetc.
![]()
Question 4.
What holds the nucleons together in a nucleus?
Answer:
The nucleons are held inside the nucleus due to the nuclear force between them which is basically a strong attractive force although the electrostatic force of repulsion between the protons is very large inside the nucleus.
Question 5.
What is the ratio of the nuclear densities of two nuclei having mass numbers in the ratio 1:3?
Answer:
Nuclear density is independent of the mass number of a nucleus. Since the two nuclei having mass numbers in the ratio 1 : 3 have the same nuclear density, so the ratio of their nuclear densities is one.
Question 6.
How the stability of a nucleus is decided?
Answer:
The stability of a nucleus is decided by its binding energy per nucleon and not by its binding energy. Higher the value of B.E. per nucleon of a nucleus greater will be its stability and vice-versa.
Question 7.
Why is the density of nucleus more than that of the atom?
Answer:
The size of the nucleus is of the order of 1(T14 m and it is made of protons and neutrons only. But the size of atom is of the order of 1010 m. Whereas the size of the atom increases by a very large factor (104 times as large as the nucleus), its mass increases only by a small amount. For this reason, the density of nucleus is very large as compared to that of the atom.
Question 8.
Why the nuclei invariably have mass less than the sum of the masses of the individual nucleons in them?
Answer:
The energy required by the nucleons to make a nucleus is provided by the nucleons at the expense of their masses. Due to this, the mass of the nucleus formed is always less than the sum of the masses of the constituent nucleons.
Question 9.
Define radioactive substance.
Answer:
It is defined as the substance whose atoms have unstable nuclei and emit α, β and γ-rays.
![]()
Question 10.
Define radioactive decay constant of a radioactive substance.
Answer:
It is defined as the reciprocal of that time in which the number of atoms of the substance become \(\frac {1}{e}\)th of the atoms present initially.
Question 11.
Why all radioactive substances seem to be identical?
Answer:
All radioactive substances seem to be identical because a radioactive sample requires infinite time to disintegrate completely.
Question 12.
Name two radioactive elements which are not found in observable quantities in nature. Why?
Answer:
Plutonium and Tritium are two radioactive elements which are not found in observable quantities in nature due to their very small values of half lives.
Question 13.
Out of a and 3 particles, which one has greater ionising power and why?
Answer:
a-particles have greater ionising power because they are massive and have large nuclear cross-section.
Question 14.
Differentiate between an electron and a P-particle.
Answer:
An electron and a p-particle are essentially the same. The electron of nuclear origin is called a P-particle. It has very large mass compared to that of an electron and travels with a high speed nearly 0.98 C.
Question 15.
Why is it not possible to define the total life of a radioactive substance?
Answer:
The radioactivity is a spontaneous process and obeys the laws of chance only. So the nucleus of an atom can have any value of total life between zero to infinity. Hence it is not possible to define the total life of a radioactive substance.
Question 16.
Define the activity of a radioactive substance.
Answer:
It is defined as the rate at which the nuclei of its atoms in the sample disintegrate. It is denoted by A or R.
∴ A or R = – \(\frac {dN}{dt}\).
Question 17.
Define the following terms :
(a) Atomic number.
(b) Mass number.
(c) Nuclear mass.
Answer:
(a) Atomic number – It is defined as the number of protons in a nucleus. It is denoted by Z. It determines the charge in the nucleus of an atom.
(b) Mass number – It is defined as the total number of protons and neutrons present in a nucleus of any atom of an element. It is denoted by A.
(c) Nuclear Mass – It is defined as the total mass of protons and neutrons present in a nucleus of an atom of any element.
Question 18.
Differentiate between endothermic and exothermic nuclear reactions.
Answer:
Endothermic Nuclear reaction – It is defined as a nuclear reaction in which the energy is absorbed during the nuclear reaction and its Q-value is negative.
Exothermic nuclear reaction – It is defined as a nuclear reaction in which the energy is evolved during the nuclear reaction and its Q- value is positive.
![]()
Question 19.
Define radioactivity.
Answer:
It is defined as the process of spontaneous emission of radiations by the disintegration of a nucleus.
Question 20.
Why only the nuclei having high atomic numbers show the process of natural radioactivity?
Answer:
In the stable nuclei having atomic mass more than 76, the number of neutrons is greater than the number of protons and thus the binding energy per nucleon decreases. Addition of neutrons in the nucleus increases the mass number but decreases the binding energy per nucleon and thus the nucleus becomes more unstable. At a certain stage no stable nuclei exist. These nuclei of high atomic number shows the phenomenon of natural radioactivity.
Question 21.
Define Curie.
Answer:
It is defined as the quantity of any radioactive substance which undergoes 3.7 x 1010 disintegrations per second. It is the S.I. unit of measuring the radioactivity of any radioactive substance.
Question 22.
Is a free neutron a stable particle?
Answer:
A neutron inside the nucleus is stable but free neutron is not a stable particle. A free neutron decays into a proton, an electron and an antineutrino \((\overline{\mathrm{v}})\) as
\({ }_{0}^{1} \mathrm{n} \rightarrow{ }_{1}^{1} \mathrm{H}+{ }_{-1}^{0} \mathrm{e}+\overline{\mathrm{v}}\).
Question 23.
Why is the energy distributing of P-radiations continuous?
Answer:
The energy distribution of P-radiations is continuous because of the emission of anti neutrino during P-decay.
Question 24.
Why electron capture is common in case of heavy nuclei?
Answer:
Electron capture is common in case of heavy nuclei because the heavy nuclei have large positive charge and small radius of K shell. Thus K shell electrons experience a large force of attraction due to positive charge of nucleus and is captured by it.
Question 25.
Neutrons produced in fission can be slowed down using ordinary water, then why is heavy water used for this purpose?
Answer:
Neutrons produced in fission have energy of the order of 2 MeV. Moderator is required to reduce this energy from 2 MeV to 0.025 eV, which is equal to the thermal motion of neutrons. This is not possible by using ordinary water to reduce energy to such an extent. Hence heavy water is used as a moderator.
![]()
Question 26.
Define nuclear reactor. Name its main components.
Answer:
Nuclear reactor – It is defined as a device in which nuclear chain reaction takes place in a controlled way.
Its main components are :
(a) Nuclear Fuel.
(b) Moderator.
(c) Control rods or Cadmium rods.
(d) Coolant.
(e) Protective shield.
Question 27.
Define Neutron reproduction factor. What are its values for different types of reactions?
Answer:
It is defined as the ratio of production of neutrons to the rate of loss of neutrons. It is denoted by k.
(a) For steady or self sustained chain reaction, k = 1.
(b) For k > 1, the fission reaction builds up fast i.e., it will be accelerated resulting in an uncontrolled and explosive situation.
(c) For k < 1, the nuclear reaction will be retarded.
Question 28.
Define Q-value of a nuclear reaction.
Answer:
It is defined as the energy equivalent to the difference in masses of the reacting nuclei and the product nuclei i.e., mathematically
Q = (mass of reactant nuclei – mass of product nuclei) x 931 MeV.
Question 29.
Define fissile material.
Answer:
It is defined as the material that can undergo nuclear fission easily.
Question 30.
Why neutrons are slowed down in a nuclear reactor?
Answer:
In case, the neutrons produced are fast, they may escape the uranium block without causing fission. When they are slowed down, then they become suitable for carrying out the nuclear reaction.
Question 31.
Define transuranic elements.
Answer:
They are defined as the elements having atomic number greater than that of Uranium i.e. 92.
Question 32.
Define nuclear fission.
Answer:
It is defined as the process of splitting of a heavy nucleus into two nuclei of comparable masses along with the emission of large amount of energy.
Question 33.
Define nuclear fusion.
Answer:
It is defined as the process in which two very light nuclei (A ≤ 8) combine to form a heavy nucleus with the liberation of large amount of energy.
![]()
Question 34.
Give one similarity and one dissimilarity between nuclear fission and fusion.
Answer:
Similarity – Both nuclear fission and fusion are similar in the sense that they are tremendous sources of energy.
Dissimilarity – The two processes are dissimilar in the sense that in fission, a heavy nucleus splits upto into two lighter nuclei while in the nuclear fusion, the two light nuclei combine to form a heavy nucleus.
Question 35.
Why nuclear fusion reactions are known as thermonuclear reactions?
Answer:
In nuclear fusion, the two light nuclei have to be brought very close to each other against the electrostatic repulsion for which the energy is made available by raising them to very high temperature of the order of 107 K. Due to this reason, nuclear fusion is called thermonuclear fusion.
Question 36.
Define thermal neutrons.
Answer:
They are defined as the neutrons having energy 0.025 eV which.corresponds to their energy at room temperature [I = \(\frac {3}{2}\) kT]
Or
They are defined as the neutrons which are being slowed down due to their collisions against the hydrogen nuclei of the moderator and cause fission of 235u nuclei.
Question 37.
Why is the neutron so effective as a bombarding particle?
Answer:
As neutron is a neutral particle, so it can penetrate the target without being repelled by the atomic nucleus or the electron cloud. The neutron increases the neutron to proton ratio of the nucleus and tends to make it unstable and hence it causes the nuclear reaction.
Question 38.
State Einstein’s mass-energy relation.
Answer:
It states that mass and energy are inter convertible according to the relation given by
E = mc2 (J)
where m is the mass, E = equivalent energy, c = speed of light.
Question 39.
Why 23892U is not suitable for chain reaction although 23892U can be readily used in chain reaction?
Answer:
The energy required to disintegrate 23892U nucleus is larger than the energy required to disintegrate 23892U nucleus. A neutron of energy 1.2 MeV is required to disintegrate 23892U nucleus. Since the neutrons of this much energy are rarely available and hence 23892U is not suitable for chain reaction. A thermal neutron of energy 0.025 eV can disintegrate 23892U and thermal neutrons are easily available. Hence 23892U can be used in chain reaction.
![]()
Question 40.
State the decay law of radioactivity.
Answer:
It states that the rate of disintegration of a radioactive substance is directly proportional to the number of atoms remained undecayed.
Question 41.
Define end point energy.
Answer:
It is defined as the maximum energy of the β-particles \(\frac { ∆L }{ L }\) emitted during P-decay of the radioactive substance.
Question 42.
Define critical mass for nuclear chain reaction.
Answer:
It is defined as the mass of the fissionable material for which the rate of production of neutrons is equal to the rate of loss of neutrons.
Question 43.
Which law is violated in the following reaction?
\(\frac { ∆L }{ L }\)
Answer:
The law of conservation of spin (i.e., angular momentum) is violated. Each particle on right side has a spin \(\frac {1}{2}\) \(\frac {h}{2π}\) so that the resultant spin on the right side is 0 or 1 (\(\frac {h}{2π}\)). But on the left side, spin is \(\frac {1}{2}\) \(\frac {h}{2π}\)
Question 44.
How are β-rays emitted from a nucleus when it does not contain electrons?
Answer:
β-rays are emitted from the nucleus due to the conversion of a neutron into a proton, an electron and antineutrino. It is this electron which is emitted in the form of β-rays and the reaction can be represented as :
![]()
Question 45.
Explain why y-decay takes place?
Answer:
γ-decay is followed by α- or β-decay. The nucleons of the nucelus are in their ground state. When the nucleus emits a or p particles, the nucleons in the daughter nucleus are raised to the higher energy states. These nucleons jump to the ground state by emitting γ- rays. The energy of a γ-ray is equal to the difference between the energies of the excited state and the ground state of the nucleons. The γ-rays are emitted by the daughter nucleus and it comes to the ground state. e.g. When 210Pb undergo β–-decay, then 210Bi is left in excited state and comes to ground state by emitting γ-rays as :
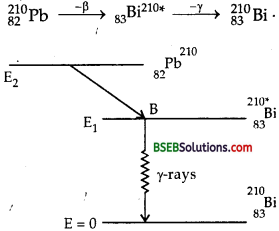
Short Answer Type Questions
Question 1.
Define Atomic mass unit. Calculate the value in kg.
Answer:
It is defined as \(\frac {1}{12}\) th of the mass of one 126C atom. It is 12 6 denoted as a.m.u. or u. It is the unit of mass commonly used in nuclear physics.
Value of lu : – We know that 6.023 x 1023 atoms of carbon-12 has mass = 12 g
∴ mass of one carbon atom = \(\frac {1}{12}\) g
∴ By definition,
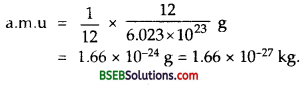
Question 2.
Express 1 u in electron volt (eV) and MeV.
Answer:
From Einstein’s mass-energy relation, we know that E = mc2
Here, m = 1 u = 1 a.m.u. = 1.66 x 10-27 kg.
c = 3 x 108 ms-1
E = 1.66 x 10-27 x (3 x 108)2
= 1.66 x 9 x 10-27 x 1016
= 1.66 x 9 x 1011 J.
1.66x9xl0-11 fc eV
1.6xl0-19
= 931.5 xlO+6eV
= 931.5 MeV (v 1 MeV = 106/eV).
![]()
Question 3.
(a) Define nuclear mass defect and derive its expression for AZX nucleus,
(b) Define and derive expression for binding energy.
Answer:
(a) Definition – It is defined as the difference between the sum of the masses of nucleons constituting a nucleus and the rest mass of the nucelus. It is denoted by ∆m.
Expression : –
Let M be the rest mass of the AZX nucleus. It contains Z protons and A-Z neutrons. Let mp and mn be the mass of a proton and a neutron.
∴ by def. ∆m = [Z mp + (A – Z) mn – M] u.
(b) It is defined as the energy equivalent to the mass defect or It is defined as the energy required to separate the nucleons infinite distance apart so that they no longer exert a force on each other.
∴ B.E. = Am. c2 (J)
= ∆m a.m.u.
= ∆m x 931.5 MeV.
Question 4.
Define average binding energy. What is its importance ?
Answer:
It is defined as the total B.E. per nucleon of a nucleus. It is denoted by \(\hat{\mathbf{i}}\) .
![]()
Its importance is that it gives a better information about the stability of the nucleus. Larger the value of g , more stable will be the nucleus and vice-versa. Thus g is more important quantity than total binding energy.
Question 5.
Draw B.E./A versus mass number (A) graph i.e., B.E. curve. What are its salient features?
Answer:
The B.E. curve is shown in the figure here.
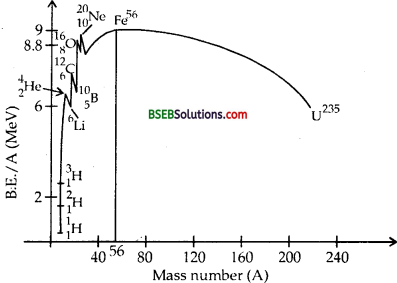
Its salient features are :
- Tine binding energy per nucleon for light nuclei such as etc. is very small.
- The B.E./nucleon increases rapidly for nuclei upto mass number 20.
- The curve has peaks corresponding to the nuclei 42He, 126C /168O and 2010Ne and these nuclei are more stable than the neighbouring nuclei.
- The B.E./A increases gradually for nuclei having mass number A > 20 and for 40 < A < 120, the curve is more or less flat.
- B.E,/nucleon in region 40 < A < 120 is about 8.5 MeV and is maximum (8.8 MeV) for 26Fe56.
- For A > 120, the B.E./nucleon starts decreasing and drops to 7.6 MeV for 23592U and is still smaller for Thus 23892is unstable and radioactive.
- This decrease in average B E is due to the increase of repulsive force among the protons when the mass number increases.
Question 6.
On the basis of the binding energy curve explain why energy should be released in the process of (a) nuclear fission, (b) nuclear fusion.
Answer:
(a) From the B.E. curve, we know that the binding energy for nucleon is smaller both for lighter and heavy nuclei. Thus when a heavy necleus splits into lighter nuclei i.e., undergo nuclear fission, we get neclei of higher value of binding energy at the cost of liberation of energy.
(b) Also when two very light nuclei combine together to form a heavier nucleus i.e., nuclear fusion takes place, the binding energy of the product nucleus is greater than the reactant nuclei, so energy is liberated. Thus during nuclear fission or fusion, the energy is liberated due to the conversion of low B.E./nucleon-nuclei into nuclei having higher value of B.E./nucleon.
Question 7.
State laws of radioactive decay.
Answer:
- The radioactivity is a spontaneous process.
- Either an α- or a β-particle is emitted during the radio active disintegration of a nucleus and both are never emitted simultaneously.
- Z and A of the daughter nucleus are reduced by 2 and 4 respectively during α-decay of the parent nucleus.
- Z of the daughter nucleus is increased by 1 and A remains same as that of the parent nucleus during the β-decay.
- Z and A of the daughter nucleus as well as of the parent nucleus remain the same during γ-decay.
- Radioactivity obeys radioactive decay law.
Question 9.
State and prove radioactive decay law.
Answer:
It states that the number of atoms disintegrated per second of a radioactive sample at any instant is always directly proportional to the number of atoms present at that time. Consider a radioactive sample having N0 atoms initially i.e., at t = 0
Let N be the no. of its atoms left undisintegrated after a time t. Also let N-dN be the number of atoms left undisintegrated at time t + dt.
∴ rate of disintegration at time t (ie., between time intervalt and t + dt) = \(\frac {dN}{dt}\)
So according to radioactive decay law.
\(\frac {dN}{dt}\) ∝N
or \(\frac {dN}{dt}\) = – λN ………(1)
where λ is the proportionality constant known as radioactive decay constant. Here negative sign shows that number of atoms present in a given element decreases as the process of disintegration continues.
∴ From eq (1), \(\frac {dN}{N}\) = -λ ……….(2)
Integrating (2), we get
\(\int \frac{\mathrm{dN}}{\mathrm{N}}=-\int \lambda \mathrm{dt}\)
or logeN = -λt + k ……..(3)
where k is the constant of integration.
To find the value of k, let us use initial conditions.
i.e., when t = O, N = N0 ………..(4)
∴ From (3) k = loge N0
∴ From (3) and (4), we get
logeN = -λt + logeN0
or logeN – loge N0 = -λt
or 1og \(\left(\frac{\mathrm{N}}{\mathrm{N}_{0}}\right)\) = -λt
or = \(\frac{\mathbf{N}}{\mathrm{N}_{0}}\) = \(e^{-\lambda t}\)
∴ N = N0\(e^{-\lambda t}\) ……….(5)
eq. (5) gives the no. of atoms left undisintegrated after a time t.
![]()
Question 10.
Define disintegration constant. How is it related to ?
Answer:
We know that
N = N0\(e^{-\lambda t}\)
Putting t = \(\frac {1}{λ}\) we get
N = N0\(\mathrm{N}_{0} \mathrm{e}^{-1}\) = \(\frac {1}{e}\) N0 = \(\frac{\mathrm{N}_{0}}{2.718}\)
= 0.368 N0.
Thus X may be defined as the reciprocal of the time after which tire original number of atoms (N0) of the given radioactive substance
reduces to \(\frac {1}{e}\) times of the original number of atoms of that substance,
We know that at t = \(\frac {T}{2}\),N = \(\frac{\mathrm{N}_{0}}{2}\)
∴ From Eq. (1), we get

i.e., Half-life time of the radioactive substance is inversely proportional to the decay constant.
Question 11.
What conclusions can you draw from the result of the density of the nucelus?
Answer:
The following conclusions can be drawn :
- As the density of nucleus is independent of the mass number of that nucleus, so the nuclear density of all the nuclei is same.
- As the density of the nucleus is of the order of 1017 kg m-3, so the nucleus is highly compact.
- The density of the nucleus is maximum at its centre and falls to zero as we move radially outwards.
Question 12.
State the properties of neutrons.
Answer:
- It is an elementary particle having no charge but its mass is 1.6748 x 1027 kg.
- It has low ionising power being neutral.
- It has high penetrating power.
- It is not deflected by electric and magnetic field.
- It can be slowed down by passing through moderators such as paraffin wax, graphite or heavy water.
- Neutron is stable inside the nucleus but unstable outside the nucleus.
- Neutron has half life period equal to 12′ and decays as follows :
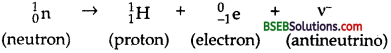
Question 13.
State properties of nuclear forces.
Answer:
- Nuclear forces are charge independent.
- Nuclear forces are always attractive in nature upto a 0.5 fermi distance and become repulsive at distances > 0.5 Fermi.
- They are non-central forces.
- They are spin dependent.
- They are exchange forces.
- They are short range forces.
- They are the strongest forces in nature.
Question 14.
A radioactive nuclide decays to form a stable nuclide. Its half life time is 3 minutes. What fraction of its 1 g will remain radioactive after 9 minutes?
Answer:
Here, \(\mathrm{T}_{1 / 2}\) = 3 minutes
∴ After 3 minutes. N = \(\frac{\mathrm{N}_{0}}{2}=\frac{1}{2}\) g
After 2 \(\mathrm{T}_{1 / 2}\), i.e., after 6 mins,
N1\(\frac{\mathrm{N}}{2}=\frac{1}{4}\) g
∴ After 9 mins i.e., in t = 3\(\mathrm{T}_{1 / 2}\) N2 = \(\frac{N_{1}}{2}=\frac{1}{2} \cdot \frac{1}{4}\) g
= \(\frac {1}{8}\) g
Question 15.
Discuss the source of energy released during the nuclear fission. Give an example.
Answer:
When 23692U is bombarded by slow neutrons, a compound nucleus 23692U is formed which disintegrates into 14156Ba and 9236Kr with the liberation of three slow neutrons and 200 MeV of energy per fission. This can be written as :
![]()
where Q = 200 MeV and is equivalent to the energy produced due to the difference in masses of nuclei before and after fission,.
Question 16.
Calculate the energy released during the fission of the nuclear reaction given in Q. 15.
Answer:
Here, mass of 23692U = 235.0439 u
mass of 10n = 1.0087 u
mass of 14156Ba = 140.139 u
mass of 9236Kr = 91.8973 u.
mass defect = ∆m
mass of (reactants – products)
= 235.0439 + 1.0087 – (140.139 + 91.8973 + 3 x 1.0087)
= 236.0526 – 235.8373
= 0.2153 a.m.u.
= 0.2153 x 931.5 MeV
= 200.4 MeV.
![]()
Question 17.
Differentiate between the nuclear fission and nuclear fusion.
Answer:
Nuclear fusion :
- Two or more lighter nuclei combine together to form a ‘ heavier nucleus.
- It requires a very high temperature of the order of 107 K.
- Very high amount of energy is liberated.
- It is used for the production of hydrogen bomb.
- Fuel (hydrogen) is cheap and easily available in plenty of amount.
- These reactions are uncontrolled.
- Fusion products are mostly non-radioactive and can be disposed off easily.
nuclear fusion :
- A heavy nucleus disintegrates into two ligher nuclei.
- It takes place at ordinary temperature.
- Low energy as compared to fusion is liberated.
- It is used for the production of nuclear bomb.
- Fuel (Uranium) is quite costly and limited in quantity.
- Controlled fission reactions are used to produce electricity.
- Fission products are generally radioactive and their disposal is a problem.
Question 18.
State various conservation laws obeyed by nuclear reactions.
Answer:
The following are the four conservation laws obeyed by a nuclear reaction:
- Law of conservation of charge – It states that in any nuclear reaction, the total charge of the nuclei or particles taking part in the nuclear reaction always remains same as the charge of the product particles or nuclei.
- Law of conservation of energy – It states that in any nuclear reaction, the total energy of the system is always conserved.
- Law of conservation of momentum – It states that in any nuclear reaction, the total momentum of atoms taking part in the nuclear reaction always remains constant.
- Conservation of mass number – It states that the total mass number always remains conserved during a nuclear reaction.
Question 19.
Distinguish between artificial and induced radioactivity.
Answer:
Artificial radioactivity – It is defined as the phenomenon of disintegration of a stable nucleus by bombarding it with a suitable high energy particle.
e.g. when Nitrogen nucleus (147N) is bombarded by an a-particle (42He)/a proton 11H is released with the formation of oxygen nucleus
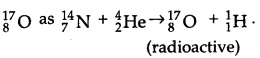
Induced radioactivity – It is the process of disintegration of a nucleus which continues even after bombarded by an a-particle 42H e.g. when 105B is bombarded by 42He, we obtain 147N which further disintegrates into 137N with the emission of a neutron and further disintegrates in 136C as follows :
105B + 42He → 147N → 137N + 10n
137N →136C + 0+1e
Question 20.
Ilustrate the successive decays of 23892U to form 23892Th 23491pa, 23492U,23490Th and 22688Ra and name the radiations emitted in each decay.
Answer:
The successive decay of 23892U takes place as :
(a) 23892U → 23490Th + 42He.
Thus radiation emitted is α-particle.
(b) 23490Th → 23491Pa + 0-1e.
∴ β-particle is emitted.
(c) 23491pa → 23492U + 0-1e
i.e., β-particle is emitted.
(d) 23491U → 23090Th + 42He
∴ α-decay take place .
(e) 23090Th →22688Ra + 42He
∴ α-decay take place .
Question 21.
Derive the expression for undecayed atoms of the radioactive substance after n half lives.
Answer:
Let N0 = original no. of atoms of a radioactive sample having half life T.
∴ After one half lifes, N = \(\frac{\mathrm{N}_{0}}{2}=\mathrm{N}_{0}\left(\frac{1}{2}\right)^{1}\)
∴ After 2 half lifes, N = \(\frac{\mathrm{N}_{0}}{4}=\mathrm{N}_{0}\left(\frac{1}{2}\right)^{2}\)
∴ After 2 half lifes, N = \(\frac{\mathrm{N}_{0}}{8}=\mathrm{N}_{0}\left(\frac{1}{2}\right)^{3}\)
Thus if N be the number of atoms left undecayed after n half lives, then
N = N0\(\left(\frac{1}{2}\right)^{n}\)
or \(\frac{N}{N_{0}}=\left(\frac{1}{2}\right)^{n}\) = \(\left(\frac{1}{2}\right)^{t / T}\) (∴t = nt)
![]()
Question 22.
Derive the relation between the activity and the Half life of a radioactive substance. .
Answer:
We know that a radioactive substance decays according to the relation,
N = N0 e-λt …………(1)
Also we know that the activity (A) of the substance is
A = \(\frac {dN}{dt}\) …………(2)
Differentiating (1) w.r.t. t, we get
\(\frac {dN}{dt}\) = N0(-λ) e-λt = -λN ………..(3)
∴ From (2) and (3), we get
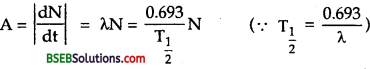
Question 23.
Calculate the K.E. of an a-particle during a-decay.
Answer:
The a-decay is represented as :
![]()
Let mα/ md be the masses of α-particle and daughter nuclei respectively.
Also let vα and vd be their respective velocities applying law of conservation of linear momentum, we get
0 = mαvα + mdvd
(∴ the parent nucleus before decay remains at rest, so px and Ex are zero.)
or vα = –\(\frac{m_{\alpha} v_{\alpha}}{m_{d}}\) ………..(1)
Also according to the law of conservation of energy.

Fro (2), and (3), we get

Question 24.
Define radiation hazard. What is the cause of radiation Hazard? Name various diseases caused.
Answer:
It is defined as the risk to the living tissues exposed to the natural radioactivity, X-rays and nuclear radiations a, p and y-rays.
The damage to the human body from nuclear radiation is caused due to the ionisation of atoms in the living cells which completely destroys the living cells.
Diseases:
(i) The radiation damage to the chromosomes in the reproductive organs can cause genetic disorder.
(ii) Long exposure to radiations causes cancer and blindness.
(iii) An acute exposure to radiations weakens or even destroys the infection resistance mechanism and may lead to death.
(iv) Radiation damage to the blood producing cells in the spleen can increase the possibility of contracting leukemia.
Question 25.
What is nuclear holocaust?
Answer:
We know that the energy released on the fission of one 23592U nucleus is about 200 MeV.
∴ Energy released due to the fission of 50 kg of 23592U
\(=\frac{200 \times 1.6 \times 10^{-13}}{0.235}\) x 6.023 x 1023 x 50 =4 x 1015 J.
which is equal to the energy produced due to 20,000 tons of T.N.T. Thus we conclude from it that the nuclear fission is a great source of energy and if 50 kg of U235 is made to undergo fission, it will result in a super explosion. In Ilnd World War, an atom bomb was dropped by USA on Hiroshima which produced an explosion equivalent to 2 x 104 tons of T.N.T. and 67 % of area of the city was destroyed and engulfed 10 km2 of the city which had population of 343,000.
About 13,5,000 people were affected and 66,000 out of it were killed. Fusion bombs are even more dangerous as the fusion reactions cannot be controlled. A hydrogen bomb may destroy every life that exists and radioactive fall out will make this planet unfit for life for all times. The radioactive waste will float like a cloud in the earth’s atmosphere. The radioactive waste will absorb the sun’s radioactives and there will be a long nuclear winter. This devastation is termed as nuclear holocaust.
Question 26.
Answer the following questions:
(a) Are the equations of nuclear reactions ‘balanced’ in the sense a chemical, equation (e.g. 2H2 + Oz -> 2HzO) is? If not, in what sense are they balanced on both sides?
(b) If both the number of protons and the number of neutrons are conserved in each nuclear reaction, in what way is mass converted into energy (or vice versa) in a nuclear reaction?
(c) A general impression is that mass-energy inter-conversion takes place only in nuclear reactions and never in chemical reactions. This is strickly speaking incorrect. Explain.
Answer:
(a) No, the equations of nuclear reactions are not balanced in the sense, a chemical reaction is balanced. In case of a chemical reaction, only the number of atoms of each element on both sides of the equation are to be balanced i.e., made same.
In a nuclear reaction, however, atomic number, mass numoer, linear momentum, angular momentum etc. has to be conserved. It is only in terms of the charge and mass number that we can balance the equations in case of nuclear reaction. ‘
![]()
(b) It is only the difference of the masses of the nuclei on the two sides of the nuclear reaction which is converted into energy. The nuclear no. in a nuclear reaction is conserved, but the total binding energy on two sides may not be equal. The difference appears as Q-value of the reaction.
(c) Mass-energy interconversion also takes place in chemical reactions also as in nuclear reactions. But the mass defect in chemical reactions is about 106 times smaller than that in nuclear reactions. Moreover in chemical reactions, the chemical energy appears in some other form of energy (say heat energy). Using relation, E = mC2, we find that the mass of products increases if their temperature is more than the reactants. Thus it is not true that mass-energy relation holds for nuclear reactions.
Question 27.
Give reasons for –
(a) Lighter elements are better moderators for a nuclear reactor than heavier elements.
(b) In a natural uranium reactor, heavy water is a preferred moderator to ordinary water.
(c) Cadmium rods are provided in a reactor.
(d) Very high temperatures as those obtained in the interior of the sun are required for fusion reaction to take place.
Answer:
(a) lighter elements are better moderator for a nuclear reactor than heavier elements because lighter elements are rich in hydrogen nuclei and when the fast moving neutrons collide against them, the neutrons almost come to rest.
(b) Neutrons produced in fission possess energy of the order of 2 MeV. Moderator is required to reduce this energy from 2 MeV to 0.025 eV which corresponds to the energy of neutrons corresponding to their thermal motion (= \(\frac {3}{2}\) KT) This is not possible by using ordinary water to reduce energy to such an extent. Hence heavy water is used as a moderator.
(c) Cadmium rods have a high cross-reaction for neutron absorption, so they are provided in a reactor. In a chain reaction, if all the neutrons produced are available take place of an uncontrolled rate. These rods absorb a suitable fraction of neutrons produced and thus a controlled chain reaction is obtained.
(d) Very high temperatures as those obtained in the interior of sun (= 107K) are required for fusion to increase thermal motion so that protons (reacting atoms) have enough kinetic energy and they come closer than the range of nuclear forces against their mutual electrostatic repulsion.
![]()
Question 28.
The energies of electrons emitted in beta decay were first measured, it was found that in the decay of some species, the electrons coming out had varying energies; the energy varied from zero to maximum. Initially the researchers felt that energy was not conserved in beta decay. If energy was conserved, they argued that, the electrons should have fixed energy, as do the alpha particles in alpha-decay. Can you explain why the argument is not true ?
Answer:
The decay of a free neutron at rest can be represented as
12n → 11H + 0-1e
According to the law of conservation of linear momentum, the e” and proton will acquire equal and opposite momentum. This means the energy of electron in the above decay is fixed in terms of the masses of the particles involved. So it is impossible for the electrons in the above decay to have a continuous distribution of energy. But if an additional particle were present, the available energy can be shared by the electron and the additional particle.
\(\frac {3}{2}\)
This simple logic was among the several arguments which led pauli to postulate the existence of third particle of zero rest mass and zero charge and is called neutrino (v). Thus” β-decay of a nucleus is represented as :
![]()
This hypothesis implies that the energy is shared by the electm and neutrino in all possible portions i.e., a low energy β-particle is accompanied by a high energy neutrino and vice-versa. Thus the total energy Q of B particle and neutrino is constant and is always equal to the end point energy. So the law of conservation of energy holds good in β-decay. It has spin = \(\frac {1}{2}\) \(\frac {n}{2π}\). It was extremely difficult to detect a neutrino because of its weak interaction with matter.
Question 29.
A free proton cannot decay into n + e+ + v, because such decay is not energetically allowed. Yet we observe in nature beta decay with positron emission. How do we understand the emission of positrons from nuclei?
Answer:
A free neutron has rest mass more than that of a proton. Thus β+ decay is energetically allowed. But the β+ decay of a free proton is not allowed energetically i.e.,
11H → 10n + 01e + v
is not allowed in case of a free proton. But in a nucleus individual neutrons and protons are not free. That is why β+ decay of proton occurs side by side. The energy needed for this decay comes from the appropriate difference in binding energies of a proton and a neutron in the neucleus.
Question 30.
The half-lives of radioactive nuclides that emit a-rays vary from microsecond to billion years. What is the reason for this large variation in the half life of a-emitters?
Answer:
We know that for nuclei having A > 140 or Z > 70, the binding energy for nucleon goes on decreasing which indicates that the stability decreases as we move towards heavier nuclei which have larger atomic number Z. They have a larger number of protons and the mutual coulomb’s repulsion of these protons results in reducing the binding effect of the nuclear forces. Inside the nucleus, the attractive nuclear forces dominate and the net potential energy is negative.
The P.E. is taken be + ve or – ve with respect to a zero level which is the energy of the a-particle when it is fa’r away from the nucleus. The a-particle bound inside the nucleus does not have enough K.E. to exceed the large Coulomb component of P.E. just outside the radius of the nucleus. There is a barrier for the particle to come out.
![]()
Gammow applied quantum physics to solve this problem and proposed that by tunneling or penetrating the barrier, a-partides can be emitted. This tunneling is directly related to the rate of disintegration i.e., activity of that sample. Also we know that the activity emitting nucleus. Thus we conclude that the a-emitters have large variation in half life due to the stability of the emitters.
Question 31.
An electron which is emitted in beta-decay comes out of the nucleus. It is not one of the atomic electrons revolving around the nucleus. This fact led some scientists to conjecture that electrons are constituents of the nucleus. Later this conjecture proved to be incorrect. Try to collect some arguments which show that this conjecture is indeed incorrect.
Answer :
The electron and a β-particle are essentially the same. The electron of nuclear origin is called a β-particle. At the time of β-decay, a neutron converts into a proton according to the following reaction :
10n →11H + 0-1e + v– In this manner, the nucleus emits electron along with antineutrino, although the nucleus does not contain the electron. Thus the conjective to think of the electron to be of nuclear origin is incorrect.
Long Answer Type Question
Question 1.
Define α, β and γ-rays. What are their properties?
Answer:
α – rays – They are defined as the helium nuclei (42He)
(helium atoms which have lost two orbital electrons). They have charge double than that of the proton and mass 4 times the mass of proton. Properties of α – particles –
- They have high ionising power.
- They have low penetration power as they are heavy particles.
- They produce heating effect when stopped and can cause skin bums.
- They slightly affect the photographic plates.
- They have rest mass four times the mass of a proton i.e., ma = 4mp
- They cause fluorescence in certain materials like ZnS, barium platinocyanide etc.
- They are deflected by electric and magnetic fields.
- α-particles are scattered by metals of high atomic weight and produce artificial disintegration in material of low atomic weight.
- They are emitted with velocities ranging between 1.4 x 107 s– 1 to 2.05 x 107 ms-1.
- They can produce artificial radioactivity in certain elements and can produce nuclear reactions.
- Their range at normal pressure in air is from 3 cm to 8 cm.
β-rays – They are the streams of electrons :
Properties of β-rays –
- They have small ionising power.
- They can affect a photographic plate.
- They are deflected by electric and magnetic fields.
- They can produce aritificial radioactivity.
- They can produce fluorescence in materials like ZnS and barium platinocyanide.
- Their range in air is several metres.
- They are deflected more when passed produce through the charged plates.
- They have average energy 2 to 3 MeV due to their extremely small mass.
- Their velocity varies from 33 % to 99.8 % of the velocity of light.
γ-rays – They are defined as the electromagnetic waves of very short wavelength of the order of 1010 to 10-12 cm.
Properties of γ-rays :
- They are highly penetrating.
- They have very small ionising power.
- They possess high energy.
- They produce fluorescence in substances like willemite.
- They can produce nuclear reactions.
- They exhibit the phenomenon of pair production.
- Their velocity is same as that of the velocity of light.
- They produce heating effect in the surface exposed to them.
- They are used in radio-therapy in hospitals.
- They knock out electrons from the surface on which they fall.
Question 2.
Find the binding energy per nucleon of nuclei 3H1, 42He and 42Li What do you infer from the values obtained?
Answer:
Here,Mass of proton, mp = 1.007825 a.m.u.
Mass of neutron, mn = 1.008665 a.m.u.
m (31H) = 3.016049 a.m.u.
m (42 He) = 4.002603 a.m.u.
m (63Li) = 6.015126 a.m.u.
(i) \(\frac{\text { B.E. }}{\text { neutron }}\) of 31H of protons = 1
number of neutrons = 3 – 1 = 2
∴ mass defect, ∆m = mp + 2mn – m(31H)
= 1.007825 + 2 x 1.008665 – 3.,016049
= 3.025155 – 3.016049 a.m.u.
B.E = ∆m x 93.15
= 0.009106 x 931.5 MeV 8.482 MeV
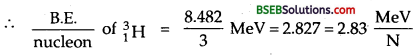
(ii) \(\frac{\text { B.E. }}{\text { nucleon of }{ }_{\mathbf{2}}^{4} \mathbf{H e}}\)
Here, no. of protons = 2
no.of neutrons = 4 – 2 = 2
= 2mp + 2mn – m (42He)
∴ mass defect, ∆m = 2 x 1.007825 +2 x 1.008665 – 4.002603
= 4.032980 – 4.002603 = 0.030377 a.m.u.
B.E. = ∆m x 931.5 MeV
= 0.030377 x 931.5 MeV = 28,2962 MeV
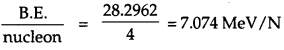
![]()
∆m = 3mp + 3mn – m(63Li)
= 3 x 1.007825 + 3 x 1.008665 – 6.015126
= 3.023475 + 3.025995 – 6.015126 a.m.u.
= 6.049470 – 6.015126 = 0.0343.44 a.m.u.
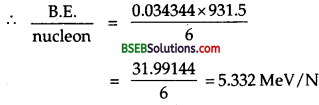
Inference from the values obtained – We conclude that :
(i) \(\frac{\text { B.E. }}{\text { nucleon }}\) for light nuclei 31H is small. 42He have mass no.4 nucleon which lies in the range 2 to 20. Thus there is sharpily defined peak corresponding to 42He which indicates that this nucleus is relatively more stable than the other nuclei in the neighbourhood.
This peak in the \(\frac{\text { B.E. }}{\text { nucleon }}\) at 42He shows a shell structure at the nuclear level.
![]()
Numerical Problems
Question 1.
Calculate : (a) the radius of 8O16 nucleus. Given R0 = 1.2 x 10-15m.
(b) the binding energy of 8O16 nucleus. Given mass of 8O16 = 15.994914 a.m.u., mp = 1.007825 a.m.u, mn = 1.008665 a.m.u.
(c) B.E./nucleon of 8O16 nucleus.
Answer:
(a) Here, R0 = 1.2 x 1015 m., A = 16
R = radius of 8O16 nucleus = ?
Using relation, R = R0 \(\mathrm{A}^{1 / 3}\), we get
R = 1.2 x 10-15 A = \(\mathrm{(16)}^{1 / 3}\) ……..(1)
Now Let r = \(\mathrm{(16)}^{1 / 3}\) ……..(2)
Taking log on both sides of eq. (2), we get
logr = \(\frac {1}{3}\) log 16 = \(\frac {1}{3}\) x 1.2041 = 0.4013
r = Antilog 0.4013 = 2.520.
or \(\mathrm{(16)}^{1 / 3}\) = 2.520 ……(3)
∴ From (1) and (3), we get
R = 1.2 x 10-15 x 2.520
= 3.024 x 10-15m.
(b) Here, p = no. of protons in 8O16 = 8.
n = no. of neutrons in 8O16 =16 – 8 = 8.
∴ mass of constituents of 8O16 nucleus = p m + n mn
= 8 x 1.007825 + 8 x 1.008665
= 8.0626 + 8.06932 = 16.13192 a.m.u.
M = mass of 8O16 = 15.994914 a.m.u.
∴∆m = mass defects of 8O16 nucleus
= 16.13192 – 15:994914 = 0.137 a.m.u.
∴B.E. of 8O16 nucleus = ∆m x 93l .5 MeV
= 0.137 x 931.5 MeV
= 127.62 MeV.
B.E. per nucleon of 8O16 nucleus = B.E./A
= \(\frac{127.62}{16}\) = 7.98 B.E./nucleon.
Question 2.
The half life of radioactive radon is 3.8 days. Find the activity of 100 g of radon. Given, atomic weight of radon = 222 and Avogadro’s number = 6.023 x 1023.
Answer:
Here, \(\mathrm{T}_{1 / 2}\) of radon = 3.8 days = 3.8 x 24 x 3,600 s.
A of Rn = 222
Number of atoms in 222 g of Radon = 6.023 x 1023.
∴ Number of atoms in 100 g of Radon = \(\frac{6.023 \times 10^{23}}{222}\) x 100
or N’ = 2.7131 x 1023
A = Activity = ?
We know that λ = \(\frac{0.693}{\mathrm{~T}_{1 / 2}}=\frac{0.693}{3.8 \times 24 \times 3,600}\) s-1
Using relation,

Question 3.
If 200 MeV energy is released in the fission of single nucleus of 23592U calculate the power output of a nuclear reactor which consumes 1 kg of 23592U Per day.
Answer:
A = Atomic weight = 235
m = mass of 23592U consuiped per day = 1 kg = 1,000 g.
E’ = energy relased per fission of 23592U = 200 MeV
= 200×1.6 x 10-13 J.
Let N’ be the number of atoms in 1 kg of 23592U .

P = Output power of nuclear reactor/day = ?
If E be the energy relased from fission of 1 kg of 23592U, then

Question 4.
The half-life of 23592U against a-decay is 1.42 x 1017 s. How many disintegrations per second occur in 1 g of 23592U? Given, N = 6.23 x 1023 mol-1.
Answer:
Here, \(\mathrm{T}_{\frac{1}{2}}\) = Half-life of 23592U = 1.42 x 1017s.
m = mass of 238U = 1 g
N = Avogadro’s no. = 6.023 x 1023.
A = Atomic weight = 238
\(\frac{\mathrm{d} \mathrm{N}}{\mathrm{d} t}\) = dismtegrations per second =?
Using relation,
\(\frac{\mathrm{d} \mathrm{N}}{\mathrm{d} t}\) = λN’ ………..(1)
Where

and N’ no. of 23592U atoms in 1 g.
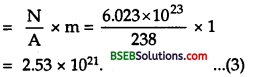
∴ From (1), (2) and (3), we get
\(\frac{\mathrm{d} \mathrm{N}}{\mathrm{d} t}\) = 4.88 x 10-18 x 2.53 x 1021
= 12.35 x 103 \(\frac{\text { dis }}{\mathrm{s}}\)
Question 5.
At a given instant, there are 25 % undecayed radio activa nuclei in a sample. After 10 s, the number of undecayed nuclei reduce to 12.5 %. Calculate : (a) mean life of the nuclei, (b) the time in whici the number of undecayed nuclei will further reduce to 6.25 % of th reduced numbçr.
Answer:
(a) Since the number of undecayed nudei is reduced frort 25 % to 12.5 % in 10 s, so half life of radioactive sample is
\(\mathrm{T}_{\frac{1}{2}}\) = 10 s
Now using the relation,
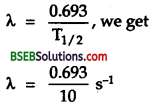
if Ta be the mean life, then
Ta = \(\frac {1}{λ}\) = \(\frac{10}{0.693}\) = 14.43 s.
(b) Let N0 = number of undecayed nuclei.
Let t = time in which N0 will reduce to 6.25 % of this number (say N).
∴ N = 6.25% N0 = \(\frac{\mathrm{N}_{0}}{16}\)
∴ Using the relation,
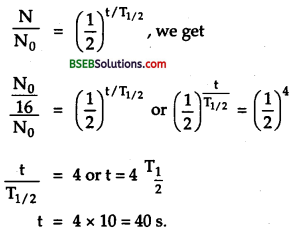
Question 6.
One gram of radium is reduced by 2 mg in 5 years by a- decay. Calculate the half period of radium. .
Answer:
Here, m0 = initial mass of Radium = 1g.
Let ∆m = mass of 23592U decayed in 5 years.
∴ t = 5 years. ∆m = 2 mg = 2 x 10-3g.
m = mass of radium left after 5 years.
∴ m = m0 – ∆m = lg – 2 x 10-3 g
= (1 – 0.002) g = 0.998 g.
\(\mathrm{T}_{\frac{1}{2}}\) = Half period of radium = ?
Using die relation,
m = m0 e-λt, we get
0.998 = 1 e-λt
eλt = \(\frac{1}{0.998}\) ………….(1)
Taking log on both sides of eq. (1), we get
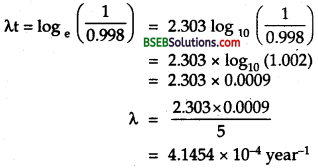
∴ Using the relation
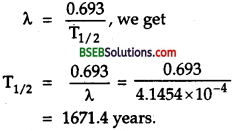
Question 7.
The half life of a radio active substance is 1672 years. If the initial mass of the substance is 1 g, after how many years will only 1 mg of it to be left behind?
Answer:
Here, m0 = initial mass of the substance = 1 g
m = 1 mg = 10-3 g
\(\mathrm{T}_{\frac{1}{2}}\) = half life period of the substance
= 1672 years,
t = ?
Using the relation,
λ = \(\frac{0.693}{\mathrm{~T}_{1 / 2}}\), we get
λ = \(\frac{0.693}{1672}\) = 4.1447 x 10-4 year-1
We know that
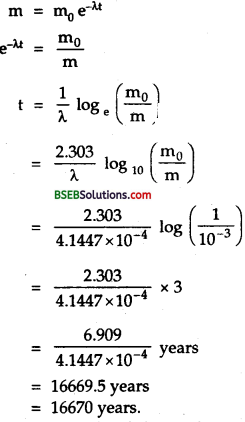
Question 8.
Calculate the energy generated in kilowatt hours, when 100 g of 73Li are converted into 42He by proton bombardment. Given mass of 73Li atom = 7.0183 amu, mass of 42He atom = 4.0040 a.m.u, mass of 11H atom = 1.0081 a.m.u.
Answer:
Here, m (73Li) = 7.0183 a.m.u.
m (42He) = 4.0040 a.m.u.
m (11H) = 1.0081 a.m.u.
The nuclear reaction taking place is given as :
73Li + 11H → 242He + Q
where Q is the Q-valu’e of die reaction given by

Also we know that 7g of 73Li contains = 6.023 x 1023 nuclei
∴n = no. of nuclei in 100 gm of 73Li = \(\frac{6.023 \times 10^{23}}{7}\) x 100
= 8.6 x 1024
Let E be the total energy released, when 100 g of 73Li is converted into 42LiHe nuclei.
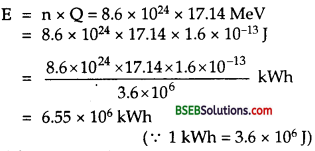
Question 9.
In a hypothetical fission reaction :
![]()
What are the values of a and b? Calculate the total energy released per nuclear fission in MeV units, Given mass of neutron = 1.009 a.m.u.
mass of X-nucleus = 235.891 a.m.u
mass of Y-riucleus = 140.673 a.m.u
mass of Z-nucleus = 91.791 a.m.u
Answer:
The nuclear reaction is :
![]()
As in a nuclear reaction both Z and A no. remain conserved.
∴ 92 = a + 36 or a = 92 – 36 = 56
and 236 = 141 + b + 3 x 1 or
b = 236 – 141 – 3 = 92.
The Q-value of the reaction is given by :
Q = (mx-m – mz – 3 mn) a.m.u.
= (235.891 – 140.673 – 91.791 – 3 x 1.009) a.m.u.
= (235.891 – 235.491) a.m.u.
= 0.4 a.m.u.
= 0.4 x 931. 5 MeV
= 372.60 MeV.
Question 10.
If 200 MeV energy is released in the fission of a single nucleus of 23592U, how many fissions must occur per second to produce a power of 1 kW?
Answer:
Here, P = 1 kW = 103 W = 103 Js-1.
Q = energy released per fission of 23592U nucleus = 200 MeV
= 200 x 1.6 x 10-13J
n = no. of fission / second = ?
Using formula,
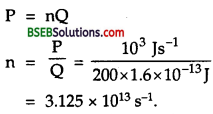
Question 11.
Some amount of a radio active substance having half life 100 days is spread inside a room and thus the level of radiation becomes 50 times the permissible level for the normal occupancy of the room. After how many days, the room will be safe for occupation?
Answer:
Here, \(\mathrm{T}_{1 / 2}\) = half-life period of substance = 100 days.
Let N be the no. of radio active atoms corresponding to the normal occupancy.
N0 = total no. of radio active atoms spread in the room.
Thus clearly as per statement,
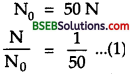
Let t = time after which the room becomes worth normal occupancy.
∴ Clearly N = no. of atoms in the room worth normal occupancy. Using the relation,

Taking logarithm on both sides of equation (2), we get

Question 12.
There is a stream of neutrons with a kinetic energy of 0.0327 eV. If the half-life of neutrons is 700 s, what fraction of neutrons will decay before they travel a distance of 10 m?
Answer:
mass of neutron = 1.675 x 10-27 kg
\(\mathrm{T}_{1 / 2}\) = half life of neutrons = 700 s
K.E. of a neutron = \(\frac{1}{2}\) mn v2 = 0.0327 eV
= 0.327 x 1.6 x 10-19J
Where V = Velocity of a neutron

S = distance covered by neutrons before decay = 10 m.
Let t = time taken by the neutron to travel 10 m.
t = \(\frac{S}{V}\) = \(\frac{10}{25 \times 10^{2}}\) = 0.4 x 10-2s
Let N0 = Total number of neutrons initially,
and N = number of neutrons left after a time t.
∴ the fraction of neutrons left after a time t is given by

∴ the fraction of neutrons decaying in a time t is given by N
1 – \(\frac{\mathrm{N}}{\mathrm{N}_{0}}\) = 1 – 0.999996 = 0.000004 = 4 x 10-6
Question 13.
It is proposed to use the nuclear reaction :
21H + 21H → 42
He in a nuclear reactor of 200 MW rating. If the energy from the above reaction is used with 25 % efficiency in the reactor, how many grams of deuterium fuel will be needed per day? The masses of 21H and 42 He nuclei are 2.0141 a.m.u. and 4.0026 a.m.u. respectively.
Answer:
The reaction and in the reactor is fusion reaction and is given by:
21H + 21H → 42He + Q
where Q-value of the reaction i.e., energy released per fusion is given by

Here, Power rating of the reactor, P = 200 MW = 200 x 106 W
t = 1 day = 24 x 3,600 s
If W be the energy to be delievered or produced per day by the reactor, then
W = P x t = 200 x 106 x 24 x 3,600 J
= 1.728 x 1013J.
n = efficiency of the reactor
= 25 % = \(\frac {25}{100}\) = \(\frac {1}{4}\)
Let W’ be the actual energy the reactor is required to produce per day.

Let W number of fusion reactions needed per day.
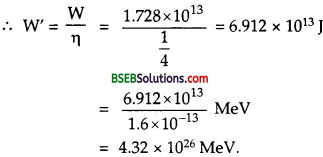
Let n = number of fusion reactions needed per day
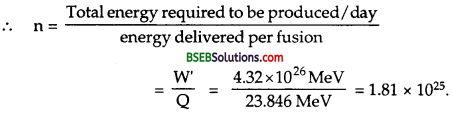
Also we know from the reaction that two deutrons are required per fusion.
∴ If N be the number of deutrons needed per day, then
N = 2 x n = 2 x 1.81 x 1025 = 3.621 x 1025.
2g of 21H contains 6.023 x 1023 deuterium nuclei. If m be the mass of deuterium needed per day then
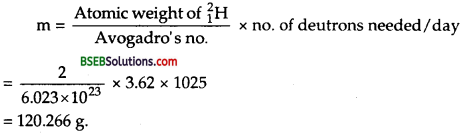
Question 14.
In a nuclear reactor 23592U undergoes fission liberating 200 MeV of energy. The reactor has 80 % efficiency and produces
1,0 MW power. If the reactor is to function for 5 years, find the total mass of uranium required initially if the reactor consumes half of its fuel in 5 years.
Answer:
Here, Power of reactor, P = 1,000 MW = 103 x 106 W = 109 W = 109 Js-1.
t = time = 5 years = 5 x 365 x 24 x 60 x 60 s = 1.577 x 108 s.
Energy delievered by the reactor in 5 years is
W = Pt = 109 x 1.577 x 108 = 1.577 x 1017J.
We know that energy generated per fission of 23592U = 200 MeV.
E = 200 x 1.6 x 10-13 J = 3.2 x 10-11 J.
n = Efficiency of the reactor = 80 % = \(\frac{80}{100}=\frac{4}{5}\)
If W’ be the actual energy, the reactor is required to produce, then using relation n = \(\frac{\mathrm{W}}{\mathrm{W}^{\prime}}\) we get
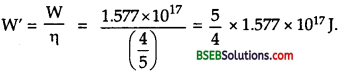
∴Total number of fissions occurred in 5 years i.e., the no. of U235 atoms required to produce needed energy given by
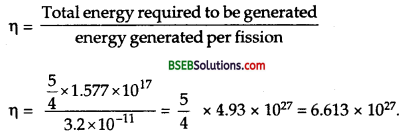
Now mass of 6.023 x 1023 nuclei of 92U235 = 235 g = 235 x 10-3 kg.
∴ Mass of 23592U consumed in five years = mass of 6.163 x 1027 nuclei
= \(\frac{235 \times 10^{-3}}{6.023 \times 10^{23}}\) x 6.163 x 1027
= 240.62 x 10 kg = 2404.62 kg.
As the reactor consumes half of its fuel in 5 years.
∴ Initial mass of 23592U required = 2 x 2404.62 kg
= 4809.24 kg
≈ 4809 kg.
Question 15.
What energy is needed to split an alpha particle to pieces? Given : mass of α-particle = 4.0039 amu, mass of proton = 1.007825 a.m.u., mass of proton = 1.008665 a.m.u. and 1 a.m.u. = 931 MeV.
Answer:
An α-particle i.e., nucleus of helium consists of two protons and two neutrons.
Here, mp = 1.007825 a.m.u.
mn – 1.008665 a.m.u. m
m(He) = 4.0039 a.m.u
Energy required to split an a-particle is equal to its binding energy.
∴ mass of protons and neutrons constituting an a-particle
= 2mp + 2mn
= 2 x 1.007825 + 2 x 1.008665
= 2.015650 + 2.017330
= 4.032980 a.m.u.
If ∆ m be the mass defect, then
∆ m = (2mp + 2mn) – m (He)
= (4.052980 – 4.0039) a.m.u.
= 0.02908 a.m.u.
∴ Energy required to split on α-particle.
= ∆m x 931 MeV
= 0.02908 x 931
= 27.07 MeV.
![]()
Question 16.
The energy of α-particles emitted from 210Po is 5.30 MeV. The half-life of this a-emitter is 138 days. Calculate: (a) the mass of 210Po needed to power a thermoelectric cell of 1 W output if the efficiency of energy conversion is 8 %? (b) the power output after 1 year.
Answer:
Here, E = energy of α-particle emitted = 5.30 MeV
Efficiency of energy conversion = q % = 8 % = \(\frac {8}{100}\), \(\mathrm{T}_{1 / 2}\) = half
life of 210Po = 138 days = 138 x 24 x 3,600 s.
P = power of thermoelectric cell = 1 W = 1\(\frac {J}{s}\)
Let n = no. of α-particles emitted per sec = number of disintegrations per second.
Let E’ be the actual energy required to be produced .
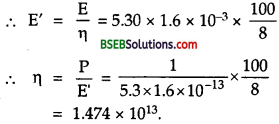
∴ A = activity = λN = 1.474 x 1013
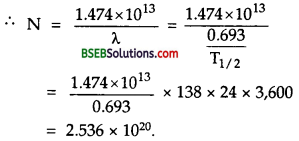
Mass of 1 atom = 210 amu = 210 x 1.66 x 10-27kg.
Mass of source = 2.536 x 1020 x 210 x 1.66 x 10-27 kg
= 88.4 x 10-6 kg = 88.4 mg.
(b) Here, t = 1 year = 365 days, P = power ouut after 1 year =?, p0 = 1 w.
We know that

Question 17.
When a neutron induces fission in a 23592U nucleus, about 185 MeV of usable energy is released. If the reactor continuously generates 100 MW of power using 23592U as fuel, then
(a) how long will it take for 2 kg of 23592U to be used up?
(b) what is the power output of the 23592U reactor if it takes 30
days to use up 2 kg of fuel and if each fission gives 185 MeV usable energy?
Answer:
(a) Here, E = energy released per fission = 185 MeV
=185 x 1.6 x 10-13 J
P = 100 MW = 102 x 106 Js-1 = 108 Js-1.
∴ no. of fissions occurring per second to give an energy output of 100 MW is given by
n = \(\frac{\mathrm{P}}{\mathrm{E}}=\frac{10^{8}}{185 \times 1.6 \times 10^{-13}}\)
= 3.378 x 1018 fissions/s.
∴ No. of nuclei contained in 2 kg of 235U
\(\frac{2 \mathrm{~kg}}{235 \times 10^{-3}}\) x 6.023 x 1023
= 2 x 2.563 x 1024 nuclei.
= 5.126 x 1024
If t be the time taken to exhaust 2 kg of 235U, then t = \(\frac{5.126 \times 10^{24}}{3.378 \times 10^{18}}\) = 17.562 days.
![]()
(b) Here, no. of atoms in 2kg of 235U
\(\frac{6.023 \times 10^{23}}{235 \times 10^{-3}}\) x 2
∴ Fission rate = \(\frac{5.126 \times 10^{24}}{30 \times 24 \times 3600}\) = 1.975 x 1018 /s.
Usable energy per fission = 185 MeV.
Power output = 185 x 1.6 x 1013 J x 1.975 x 1018/s
= 58.46 x 106W
= 58.46 MW.
Question 18.
The radius of nucleus with nucleon number 15 is 3 x 1015 m. Find the radius of nucleus with nucleon number 120.
Answer:
Here, R1= 3x 10-15 m.
A1 = 15
A1 = 120.
R2 = ?
We know that the radius of nucleus is given by


Question 19.
A nucleus ruptures into two nuclear parts which have their velocities ratio equal to 2 : 1. What will be the ratio of their nuclear size?
Answer:
Let mx and m1 be m2the masses of two nuclear parts having velocities v1 and v2 respectively, v1: v2 = 2 : 1
Before rupture, the nucleus remains at rest, so according to the law of conservation of momentum,
0 = m1v1 + m1v2
or \(\frac{\mathrm{m}_{1}}{\mathrm{~m}_{2}}=-\frac{\mathrm{v}_{2}}{\mathrm{v}_{1}}\) = 1 : 2
As the radius of nucleus is given by R = R0 \(A^{\frac{1}{3}}\)
where A = mass no.
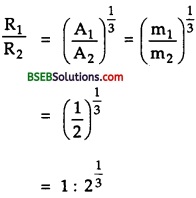
Question 20.
A star has 1040 deutrons. It produces energy via the process :
21H + 21H → 31H + P and 21H + 31H → 42He + n
If the average power radiated by star is 1016 watt, find the time in which deutron supply would be enhausted. The masses of the nucleus are : m ![]() = 2.014 a.m.u, m (p) 1.007 a.m.u, m (n) = 1.008 a.m.u. and m (4He) = 4.001 a.m.u.
= 2.014 a.m.u, m (p) 1.007 a.m.u, m (n) = 1.008 a.m.u. and m (4He) = 4.001 a.m.u.
Answer:
Adding two reactions, we get
321H -> 42He + 11P + 11n
∆m = 3m (21H) – m (42 He)
-m(21P) – m (10n)
= 0.0026 a.m.u.
Energy produced due to fusion of 3 (21H) = 0.0026 x 931 MeV
= 0.0026 x 931 x 106 x 1.6 x 10-19 J.
Eenergy produced by 1040 deutrons = \(\frac{10^{40}}{3}\) x 26 x 931 x 1.6 x 10-17
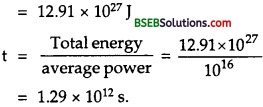
Question 21.
Calculate the mass of one Curie of U235.
Answer:
We know that 1 Curie = 3.7 x 1010 dps.
∴Number of atoms in 1 Curie = 3.7 x 1010
Mass of 6.023 x 1023 atoms of Uc = 235 g
∴Mass of 3.7 x 1010 atoms of U235 = \(\frac{235}{6.023 \times 10^{23}}\) x 3.7 x 1010
= 144.36 x 10-13 g = 14.44 x 1010-12
Question 22.
Obtain the approximate value of the radius of (a) a nucleus of. 4 He and (b) a nucleus of 23592U (c) What is the ratio of these radii? (Assume R0 = 1.2 x 10-15 m.)
Answer:
We know that
R = R0 \(A^{\frac{1}{3}}\) ……….(1)
Where R0 = 1.2 x 10-15 m.
![]()
(a) Radius of 42He nucleus :
Here, A = 4
Let R1 be the radius of 42He nucleus.
∴ From (1), we get
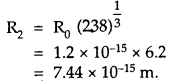
(b) Radius of 23592 U :
Here, A = 238
If Rj be the radius of 23592 U nucleus, then
Using equation (1), we get
R 2 = R0 \((238)^{\frac{1}{3}}\)
= 1.2 x 10-15 x 6.2
= 7.44 x 10-15 m.
(c) The ratio of there radii is given by,
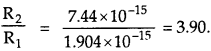
Aliter: (a) R1 = 1.2 x 10-15 x \((4)^{\frac{1}{3}}\)
= 10-15 x 1.587 m
= 1.59 x 10-15m.
R2 = 1.2 x 10-15 x \((238)^{\frac{1}{3}}\)
= 10-15 x 6.2m
= 6.2 x 10-15 m.
Fill In The Blanks
Question 1.
Atoms having the same …………… but different ………….. are called isotopes.
Answer:
Atomic number, mass number.
Question 2.
The size of the nucleus varies as …………. power of mass number.
Answer:
\(\frac {1}{3}\)
Question 3.
…………. is the process in which light nuclei fuse together to form a heavy nucleus.
Answer:
Nuclear fusion.
Question 4.
In the fission of 23592U nucleus on an average …………. neutrons are released.
Answer:
2.5.
Question 5.
In a nuclear reactor, heavy water is used as a …………. which slows down the neutrons.
Answer:
Moderator.
Question 6.
The difference between 23592U and 23592U is that 23592U contains three more …………. and fission of 23592U is caused by …………. neutrons while fission of 23592U is caused by …………. neutrons.
Answer:
neutrons, fast, slow.
Question 7.
The order of magnitude of the density of nuclear matter is ………….
Answer:
1017 kg m-3.
![]()
Question 8.
The average binding energy per nucleon for the nuclei lying in the middle of periodic table is nearly ………….
Answer:
8.5 MeV.
Question 9.
Two deuterium nuclei can combine to form a …………. if they possess sufficiently ………….
Answer:
Helium nucleus, high kinetic energy.
Question 10.
The average energy released per fission of 23592U s approximately …………. MeV.
Answer:
200.
Question 11.
Proton was discovered by …………. and neutron was discovered by ………….
Answer:
Rutherford, Chadwick.
Question 12.
The ratio of the radii of the nuclei 13A127 and 52Te125 is approximately ………….
Answer:
3:5.
Question 13.
…………. was the fissionable material used in the bomb dropped at Nagasaki in 1945.
Answer:
Plutonium.
Question 14.
An element A decays into element C by a two step process:
A → B + 42He
B → C + 2e–
Then A and C are ………….
Answer:
Isotopes.
Question 15.
The sun obtains its radiant energy from ………….
Answer:
Fusion process.
Question 16.
The B.E. per nucleon is maximum for ………….
Answer:
5626Fe.
![]()
Question 17.
Slow neutrons are incident on a sample of uranium containing both 23592U and 23592U isotopes, then only …………. atoms will undergo fission.
Answer:
23592U
Question 18.
The critical mass of the fissionable material is ………….
Answer:
The minimum mass needed for chain reaction.
Question 19.
The bulk of energy released in nuclear fission process appears as ………….
Answer:
Kinetic energy of fission fragments.
Question 20.
In a given reascion :
![]()
the radioactive radiations are emitted in.the sequence of ………….
Answer:
α, β, γ
Question 21.
A positron has the same mass as ………….
Answer:
electron.
Question 22.
Neutrino is a particle with ………….
Answer:
Chargeless property and has spin.
Question 23.
Out of α, β and γ radiations, …………. and …………. are affected by a magnetic field.
Answer:
Alpha (α) and.beta (β) radiations.
![]()