BSEB Bihar Board Class 10 Science Solutions Chapter 2 Acids, Bases and Salts
Bihar Board Class 10 Science Solutions Chapter 2 Acids, Bases and Salts Textbook Questions and Answers, Additional Important Questions, Notes.
Bihar Board Class 10 Science Chapter 2 Acids, Bases and Salts InText Questions and Answers
Intext Question (Page 18)
Question 1.
You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic and a basic solution, respectively. If you are given only red litmus paper, how will you identify , the contents of each test tube ?
Answer:
First of all, we will put one strip of red litmus paper in each test tube. In one of the test tubes, the litmus paper will turn t blue. It is sure that this test tube contains basic solution. The , remaining two test tubes contain distilled water and acidic solution in both of which red litmus remains red.
Now, we will pour out some basic solution from the test tube so that only a little amount remains in it. Then, we will pour the contents of one of the remaining test tubes into the test tube containing basic solution. Two things may happen.
1. The litmus may turn red again : This indicates that the second test tube contains acidic substance. And the other one contains distilled water.
2. The litmus may remain blue : This indicates that the poured liquid is distilled water. And .the other one is the acidic solution.
![]()
Intext Questions (Page 22)
Question 1.
Why should curd and sour substances not be kept in brass and copper vessels ?
Answer:
Curd and sour substances contain acids. Acids react with metals to give salt and hydrogen gas. So, if such substances are kept in copper container, the acid will react and the container will be corroded.
Question 2.
Which gas is usually liberated when an acid reacts with a metal ? Illustrate with an example. How will you test the presence of this gas ?
Answer:
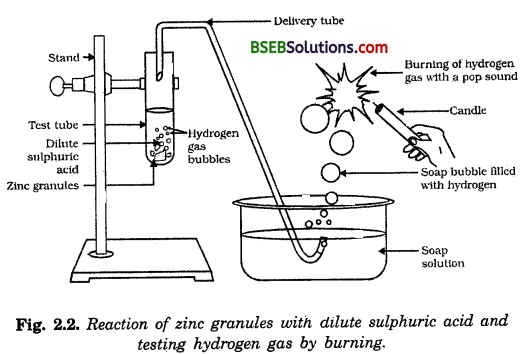
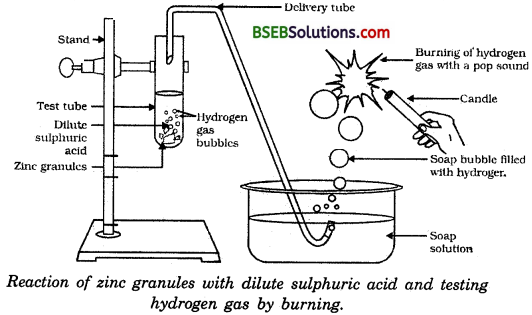
Usually, hydrogen gas is liberated when an acid reacts witn a metal. Let us take the reaction between zinc and sulphuric acid.
The apparatus is set as shown in Fig. 2.2. About 5 ml of dil. H2S04 is taken in the test tube and a few. granules of zinc are added to it. The delivery tube is caped in the test tube so that the evolved gas passes through the soap solution.
Soap bubbles filled with the evolved gas come out of the solution. The flame of a lighted candle is brought in contact with the bubble. The gas in the bubble burns with a pop sound. This proves that the gas evolved in the reaction between zinc and sulphuric acid is hydrogen.
Question 3.
A metal compound reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one «f the compounds formed is calcium chloride.
Answer:
CaCO3 + 2HCl -> CaCl2 + H2O + CO2
![]()
Intext Questions (Page 25)
Question 1.
Why do HCl, HNO3, etc. show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character ?
Answer:
HCl, HNO3, etc. have releasable H+ ion. When dissolved in water, the H+ ion gets sepaVated and shows acidic character. In alcohol or glucose, there is no releasable H+ ion.
Question 2.
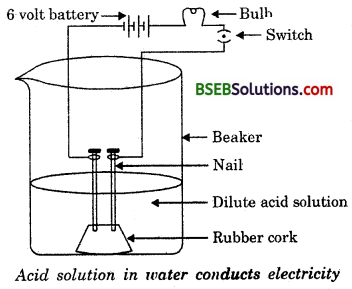
Why does an aqueous solution of an acid conduct electricity ?
Answer:
When dissolved in water, acids dissociate to form ions
e-g., HCl + H2O → Cl– + H3O+
These ions are responsible for electrical conductivity.
Question 3.
Why does dry HCl gas not change the colour of the dry litmus paper ?
Answer:
Dry HCl does not dissociate to give H+ ions. So, it does not show acidic character.
Question 4.
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid ?
Answer:
The process of dissolving an acid or a base in water is a highly’ exothermic one. Care must be taken while mixing concentrated nitric acid or sulphuric acid with water. The acid must always be added slowly to water with constant stirring. If water is added to a concentrated acid, the heat generated may cause the mixture to splash out and cause bums. The glass container may also break due to excessive local heating.
![]()
Question 5.
How is the concentration of hydronium ions (H3O+) affected when a solutipn of an acid is diluted ?
Answer:
Concentration of H3O+ ion decreases.
Question 6.
How is the concentration of hydroxide ions (OH–) affected when excess base is dissolved in a solution of sodium hydroxide ?
Answer:
Concentration of OP- group increases.
In text Questions (Page 28)
Question 1.
You have two solutions, A and B. The pH of solution A is -6 and pH of solution B is 8. Which solution has more hydrogen ion concentration ? Which of this is acidic and which one is basic ?
Answer:
Solution A has more H+ ion concentration. Solution A is acidic and solution B is basic.
Question 2.
What effect does the concen-tration of H+(ag) ions have on the nature of the solution ?
Answer:
As the concentration of H+ ions increases the solution becomes more acidic.
Question 3.
Do basic solutions also have H+(aq) ions ? If yes, then why are these basic ?
Answer:
This H+ ion comes from water.
![]()
Question 4.
Under what soil condition do you think a farmer would treat the soil of his fields with quick lime (calcium oxide) or slaked lime (calcium hydroxide) or chalk (calcium carbonate) ?
Answer:
If the farmer finds his soil turned acidic, he should use the bases to neutralise it.
Intext Questions (Page 33)
Question 1.
What is the common name of the compound CaOCl2?
Answer:
Bleaching powder.
Question 2.
Name the substance which on treatment with chlorine yields bleaching powder.
Answer:
Dry slaked lime [Ca(OH)2].
Question 3.
Name the sodium compound which is used for softening hard water.
Answer:
Washing soda i.e., sodium carbonate (Na2CO3).
Question 4.
What will happen if a solution of sodium – hydrocarbonate is heated ? Give the equation of the reaction involved.
Answer:
The following reaction takes place when it is heated during cooking :
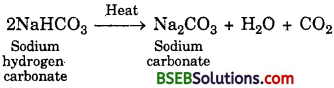
Question 5.
Write an equation to show the reaction between Plaster of Paris and water.
Answer:
CaSO4.\(\frac {1}{2}\)H2O + 1\(\frac {1}{2}\)H2O → CaSO4.2H2O
Bihar Board Class 10 Science Chapter 2 Acids, Bases and Salts Textbook Questions and Answers
Question 1.
A solution turns red litmus blue, its pH is likely to be –
(a) 1
(b) 4
(c) 5
(d) 10
Answer:
(d) 10
Question 2.
A solution reacts with crushed egg-shells to give a gas that turns lime-water milky. The solution contains –
(a) NaCl
(b) HCl
(c) LiCl
(d) KCl
Answer:
(6) HCl
![]()
Question 3.
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount HCl solution (the same solution as before) required to neutralise it will be –
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
Answer:
(d) 16 mL
Question 4.
Which one of the following types of medicines is used for treating indigestion ?
(a) Antibiotic
(b) Analgesic
(c) Antacid
(d) Antiseptic
Answer:
(c) Antacid
Question 5.
Write word equations and then balanced equations for the reaction taking place when :
(а) dilute sulphuric acid reacts with zinc granules.
(b) dilute hydrochloric acid reacts with magnesium ribbon.
(c) dilute sulphuric acid reacts with aluminium powder.
(d) dilute hydrochloric acid reacts with iron filings.
Answer:
(a) dil. Sulphuric acid + Zinc → Zinc sulphate + Hydrogen gas
H2SO4(aq) + Zn ZnSO4(aq) + H2(g)
(b) dil. Hydrochloric acid + Magnesium → Magnesium
chloride + Hydrogen gas 2HCl(aq) + Mg → MgCl2(aq) + H2(g)
(c) dil. Sulphuric acid + Aluminium → Aluminium sulphate + Hydrogen gas
3H2SO4(aq) + 2Al(s) → Al2(SO4)3(aq) + 3H2(g)
(d) dil. Hydrochloric acid + Iron → Ferrous chloride + Hydrogen gas
6HCl(aq) + 3Fe(s) → 3FeCl2(ag) + 3H2(g)
![]()
Question 6.
Compounds such as alcohols and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
Answer:
The apparatus is set as shown in figure. Solutions of glucose is added and the switch is turned on. The bulb does not glow. This means glucose does not dissociate into ions on dissociation. So, glucose is not an acid. Similarly, alcohol is also not an acid.switch Acid solution in water conducts electricity.
Question 7.
Why does distilled water not conduct electricity, whereas rain water does ?
Answer:
Distilled water does not dissociate into ions. So, it does not conduct electricity. Rain water has dissolved CO2 in it which becomes carbonic acid.
CO2 + H2O → H2CO3
This carbonic acid dissociate into ions.
H2CO2 + 2H2O (H3O+)2 + CO3-2
These ions are responsible for electrical conductivity of rain water.
Question 8.
Why do acids not show acidic behaviour in the absence of water ?
Answer:
In absence of water, acids do not dissociate. So, they do not show acidic behaviour.
Question 9.
Five solutions A, B, C, D and E when tested with universal indicator showed pH as 4, 1, 11, 7 and 9, respectively. Which solution is :
(a) neutral ?
(b) strongly alkaline ?
(c) strongly acidic ?
(d) weakly acidic ?
(e) weakly alkaline ?
Arrange the pH in increasing order of hydrogen-ion concentration.
Answer:
(a) D, (b) C, (c) B, (d) A, (e) E.
Increasing order of H+ concentration :
C (11) < E (9) < D (7) < A (4) < B (1).
Question 10.
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. In which test tube will the fizzing occur more vigorously and why ?
Answer:
In test tube A, fizzing occur more vigorously because HCl is a strong acid and dissociate more.
Question 11.
Fresh milk has a pH of 6. How do you think the pH will change as it turns into curd ? Explain your answer.
Answer:
Curd is sour which means its pH will decrease from 6.
![]()
Question 12.
A milkman adds a very small amount of baking soda to fresh milk.
(a) Why does he shift the pH of the fresh milk from 6 to slightly alkaline ?
(b) Why does this milk take a long time to set as curd ?
Answer:
(a) Baking soda is alkaline which causes the alkalinity of milk.
(b) Curd needs acidic condition-to set.
Question 13.
Plaster of Paris should be stored in a moisture- proof container. Explain why?
Answer:
Plaster of Paris easily absorbs water and forms hard gypsum. Thus, if Plaster of Paris is not kept in a moisture-proof container, then all Plaster of Paris will get converted into gypsum.
Question 14.
What is a neutralisation reaction ? Give two examples.
Answer:
The reaction between an acid and a base to give a salt and water is known as a neutralisation reaction.
NaOH(ag) + HCl(ag) → NaCl(ag) + H2O(l)
Ca(OH)2(ag) + H2SO4(aq) → CaSO4 + H2O
Question 15.
Give two important uses of washing soda and baking soda.
Answer:
Uses of washing soda :
(i) Sodium carbonate (washing soda) is used in glass, soap and paper industries.
(ii) It is used in the manufacture of sodium compounds such as borax.
Uses of baking soda :
(i) Sodium hydrogencarbonate is also an ingredient in antacids. Being alkaline, it neutralises excess acid in the stomach and provides relief.
(ii) It is also used in soda-acid fire extinguishers.
Bihar Board Class 10 Science Chapter 2 Acids, Bases and Salts Textbook Activities
Activity 2.1 (Page 18)
Question 1.
Tabulate your observation in Table 2.1.
Answer:

Activity 2.2 (Page 18)
Question 1.
Test the change of odour of clove oil, vanilla with dil. HCl and dilute NaOH.
Answer:

Answer:
Do it yourself.
Activity 2.3 (Page 19)
Question 1.
What do you observe on the surface of the zinc granules ?
Answer:
Bubble of a gas.
Question 2.
Why are bubbles formed in the soap solution ?
Answer:
The gas evolved, in the reaction between zinc and sulphuric acid passes through the delivery tube into the soap solution. Due to this, bubbles are formed.
![]()
Question 3.
What do you observe ?
Answer:
The bubble bursts and the gas inside it burns with a pop sound.
Question 4.
Are the observations in all the cases the same or different ?
Answer:
Same.
Activity 2.4 (Page 20)
Question 1.
Record your observations.
Answer:
(i) Bubbles of a gas are formed on the surface of zinc granules.
(ii) The bubbles burst and gas inside them burns with pop sound, when a lighted candle is brought near them.
Activity 2.5 (Page 20)
Question 1.
What do you observe ?
Answer:
A gas is formed.
Question 2.
Record your observation.
Answer:
The lime water turns milky.
Activity 2.6 (Page-21)
Question 1.
What is the colour of the solution ?
Answer:
Pink.
Question 2.
1s there any colour change for the reaction mixture ?
Answer:
Yes, it gradually becomes colourless.
Question 3.
Why did the colour of phenolphthalein change after the addition of an acid ?
Answer:
Acid nullifies the effect of base.
Question 4.
Does the pink colour of phenolphthalein reappear ?
Answer:
Yes.
Question 5.
Why do you think this happened ?
Answer:
By adding. NaCl, the solution again becomes basic.
![]()
Activity 2.7 (Page 21)
Question 1.
What has happened to the copper oxide ?
Answer:
Copper oxide reacts with HCl to form CuCl2.
Activity 2.8 (Page 22)
Question 1.
What do you observe ?
Answer:
Dilute nitric acid and dilute sulphuric acid conduct electricity.
Question 2.
Repeat the experiment separately with glucose and alcohol solutions. What do you observe now?
Answer:
Glucose does not conduct electricity. Alcohol does not conduct electricity.
Question 3.
Does the bulb glow in all cases?
Answer:
No.
Activity 2.9 (Page 23)
Question 1.
What do you observe? Is there a gas coming out of the delivery tube?
Answer:
Bubbles form when sulphuric acid is added. Yes, there is a gas coming out of the delivery tube.
Question 2.
In which case does the litmus paper change colour ?
Answer:
Moist litmus changes the colour (blue to red).
Question 3.
On the basis of the above activity, what do you infer about the acidic character of :
(i) dry HCl gas
(ii) HCl solution ?
Answer:
(i) Dry HCl gas does not show acidic character.,
(ii) HCl solution shows acidic character.
Activity 2.10 (Page 24)
Question 1.
Is there a change in temperature ?
Answer:
Yes.
Question 2.
Is this an exothermic or endothermic process ?
Answer:
Exothermic.
![]()
Activity 2.11 (Page 26)
Question 1.
Record your observations.
Answer:
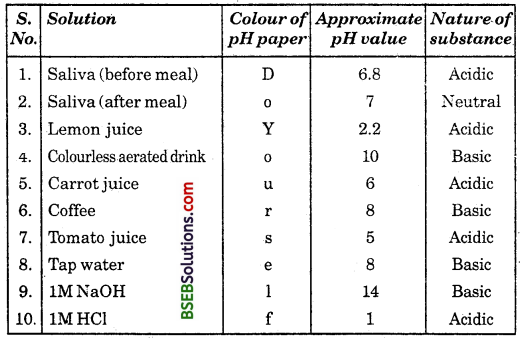
Question 2.
What is the nature of each substance on the basis of your observation?
Answer:
The nature of each substance is given in the above table.
Activity 2.12 (Page 27)
Question 1.
What can you conclude about the ideal soil pH for the growth of plants in your region ?
Answer:
Ideal pH range for soil is 7 to 7.6.
Activity 2.13 (Pages 28-29)
Question 1.
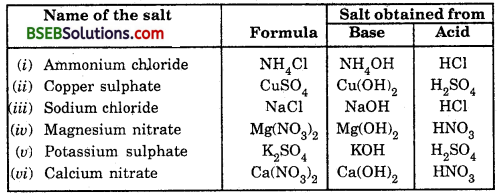
Write the formulae of the salts given below : Potassium sulphate, sodium sulphate, calcium sulphate, magnesium sulphate, copper sulphate, sodium chloride, sodium nitrate, sodium carbonate and ammonium chloride.
Answer:
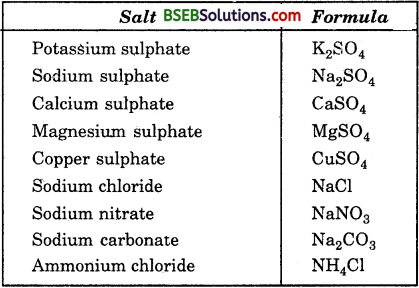
Question 2.
Identify the acids and bases from which the above salts may be obtained.
Answer:
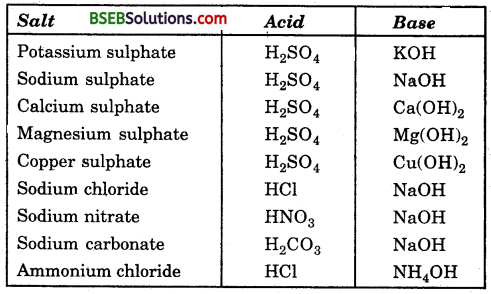
Question 3.
How many families can you identify among the salts given in this activity ?
Answer:
Sulphate salts : K2SO4, Na2SO4, CaSO4, MgSO4, CuSO4.
Chloride salts : NaCl, NH4Cl.
Sodium salts : Na2SO4, NaCl, Na2CO3, NaNO3.
![]()
Activity 2.14 (Page 29)
Question 1.
Report your observations in Table 2.4.
Answer:
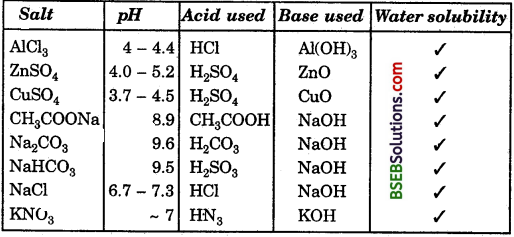
Question 2.
Check all of above of these solutions on litmus which of the salts are acidic, basic or neutral ?
Answer:
Acidic – Aluminium chloride, zinc sulphate, copper, sulphate.
Basic – Sodium acetate, sodium carbonate, sodium, hydrogencarbonate.
Neutral – Sodium chloride, sodium nitrate.
Activity 2.15 (Page 32)
Question 1.
What is the colour of the copper sulphate after .. heating ? s
Answer:
White.
Question 2.
Do you notice water droplets in the boiling tube ?
Where have these come from ?
Answer:
Yes, they come from the CuSO4 crystal.
Question 3.
What do you observe ? Is the blue colour of copper sulphate restored ?
Answer:
On adding water, blue colour of CuSO4 crystal is restored.
Bihar Board Class 10 Science Chapter 2 Acids, Bases and Salts NCERT Exemplar Problems
Short Answer Type Question
Question 1.
Match the acids given in Column (A) with their correct source given in Column (B).
| Column (A) | Column (B) |
| (а) Lactic acid | (i) Tomato |
| (b) Acetic acid | (ii) Lemon |
| (c) Citric acid | (iii) Vinegar |
| (d) Oxalic acid | (iv) Curd |
Answer:
(a) → (iv)
(b) → (iiii)
(c) → (ii)
(d) → (i)
Question 2.
Match the important chemicals given in Column (A) with the chemical formulae given in Column (B).
| Column (A) | Column (B) |
| (a) Plaster of Paris | (i) Ca(OH)2 |
| (b) Gypsum | (ii) CaSO4.V6 H2O |
| (c) Bleaching Powder | (iii) CaSO4.2H2O |
| (d) Slaked Lime | (iv) CaOCl |
Answer:
(a) → (ii)
(b) → (iii)
(c) → (iv)
(d) → (i)
Question 3.
What will be the action of the following substances on litmus paper?
Dry HCl gas, Moistened NH3 gas, Lemon juice, Carbonated soft drink, Curd, Soap solution.
Answer:

Question 4.
Name the acid present in ant sting and give its chemical formula. Also give the common method to get relief from the discomfort caused by the ant sting.
Answer:
The acid present in ant sting is methanoic acid (formic acid). The chemical formula is HCOOH. To get relief one should apply any available basic salt e.g., baking soda (NaHCO3) on it.
![]()
Question 5.
What happens when nitric acid is added to egg shell?
Answer:
Egg shells contain calcium carbonate. When nitric acid is added to it, carbon dioxide gas is evolved.
The reaction can be given as –
CaCO3+ 2HNO2 – Ca(NO3)2 + H2O + CO2
Question 6.
A student prepared solutions of (i) an acid and (ii) a base in two separate beakers. She forgot to label the solutions and litmus paper is not available in the laboratory. Since both the solutions are colourless, how will she distinguish between the two?
Answer:
She can distinguish between the solutions of an acid and a base by testing their samples with chemical indicators like phenolphthaiein or natural indicators like turmeric or china rose. If she adds few drops of phenolphthaiein to the samples of both the solutions, the sample in which the colour of phenolphthaiein turns pink is a base and the other is acid.
Question 7.
How would you distinguish between baking powder and washing soda by heating?
Answer:
The chemical formula of baking powder is sodium hydrogencarbonate (NaHCO3). Whereas,-that of washing soda is sodium carbonate (Na2CO3.10H2O). Sodium hydrogencarbonate on heating gives CO2 gas which will turn lime water milky whereas no such gas is obtained from sodium carbonate.
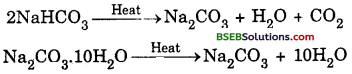
Question 8.
Salt A commonly used in bakery products on heating gets converted into another salt B which itself is used for removal of hardness of water and a gas C is evolved. The gas C when passed through lime water, turns it milky. Identify A, B and C.
Answer:
Baking powder (NaHCO3), salt A is commonly used in bakery products. On heating it forms sodium carbonate (Na2CO3), B and CO2 gas, C is evolved. When CO2 gas is passed through lime water it forms calcium carbonate (CaCO3), which is slightly soluble in water making it milky.
A – NaHCO3
B – Na2CO3
C – CO2 gas
Question 9.
In one of the industrial processes used for manufacture of sodium hydroxide, a gas X is formed, as by product. The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in chemical industry. Identify X and Y giving the chemical equation of the reactions involved.
Answer:
In the manufacture of sodium hydroxide, hydrogen gas and chlorine gas (X) are formed as by-products. When chlorine gas (X) reacts with lime water, it forms calcium oxychloride (bleaching powder) Y.
The reactions are –
2NaCl(aq) + H2O(l) → 2NaOH(aq) + Cl2(g) + H2(g)
X → Cl2 (Chlorine gas)
Ca(OH)2(s) + Cl2(g) → CaOCl2(s) + H2O
Y → Calcium oxychloride (bleaching powder)
Question 10.
Fill in the missing data in the following table

Answer:
Question 11.
What are strong and weak acids? In the following list of acids, separate strong acids from weak acids:
Hydrochloric acid, citric acid, acetic acid, nitric acid, formic acid, sulphuric acid.
Answer:
In aqueous solutions, strong acids ipnise completely and provide hydronium ions. On the other hand, weak acids are partially ionised and an aqueous solution of same molar concentration provides a much smaller concentration of HgO+ ions.
Strong acids – Hydrochloric acid, sulphuric acid, nitric acid
Weak acids – Citric acid, acetic acid, formic acid
![]()
Question 12.
When zinc metal is treated with a dilute solution of a strong acid, a gas is evolved, which is utilised in the hydrogenation of oil. Name the gas evolved. Write the chemical equation of the reaction involved and also write a test to detect the gas formed.
Answer:
When zinc reacts with dilute solution of strong acid, it forms salt and hydrogen gas is evolved.
Zn + 2HCl → ZnCl2 + H2
When a burning splinter is brought near the mouth of the test tube, the gas burns with a pop sound.
Long Answer Type Questions
Question 13.
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure, what would happen if following changes are made :
(a) In place of zinc granules, same amount of zinc dust is taken in the test tube ?
(b) Instead of dilute sulphuric acid, dilute hydrochloric acid is taken ?
(c) In place of zinc, copper turnings are taken ?
(d) Sodium hydroxide is taken in place of dilute sulphuric acid and the tube is heated.
Answer:
(a) If in place of zinc granules, same amount of zinc dust is taken in the test tube, hydrogen gas will evolve with greater speed. It is so because dil. H2SO4 will get more surface area in contact to react in case of zinc dust than in case of zinc granules.
(b) If instead of dilute sulphuric acid, dilute hydrochloric acid is taken, almost same amount of hydrogen gas is evolved. It happens so because zinc reacts with dil. HCl in the same manner as it reacts with dil. H2SO4.
(c) If copper turnings are taken at the place of zinc, hydrogen gas is not evolved as dil. H2SO4 reacts differently with Zn and Cu.
(d) If sodium hydroxide is taken in place of dilute sulphuric acid and tube is heated hydrogen gas will be evolved. The following reaction would take place :
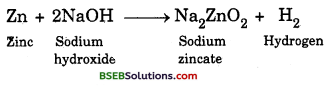
Question 14.
For making cake, baking powder is taken. If at home your mother uses baking soda instead of baking powder in cake,
(a) how will it affect the taste of the cake and why?
(b) how can baking soda be converted into baking powder?
(c) what is the role of tartaric acid added to baking soda?
Answer:
(a) Baking soda is sodium hydrogencarbonate. On heating, it is converted into sodium carbonate which is bitter to taste
![]()
Thus, if baking soda is taken in place of baking powder in cake, it would taste bitter.
(b) Baking soda can be converted into baking powder by the addition of appropriate amount of tartaric acid to it.
(c) The role of tartaric acid is to neutralise sodium carbonate and hence to prevent cake from gettting bitter in taste.
![]()
Question 15.
A metal carbonate X on reacting with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, it gives a compound Z, used for disinfecting drinking water. Identify X, Y, G and Z.
Answer:
The gas evolved at anode during electrolysis of brine is chlorine (G). When chlorine gas is passed through dry Ca(OH)2 (Y) produces bleaching powder (Z) used for disinfecting drinking water.

Since Y and Z are calcium salts, therefore X is also a calcium salt and is calcium carbonate.
CaCO3 + 2HCl → CaCl2 + CO2+ H2O
Ca(OH)2 + CO2 → CaCO3 + H2O
Question 16.
A dry pellet of a common base B, when kept in open absorbs moisture and turns sticky. The compound, is also a by-produpt of chloralkali process. Identify B. What type of reaction occurs when B is treated with an acidic oxide? Write a balanced chemical equation for one such solution.
Answer:
Sodium hydroxide (NaOH) is a commonly used base and is hygroscopic, that is, it absorbs moisture from the atmosphere and becomes sticky. The acidic oxides react with base to give salt and water. The reaction between NaOH and CO2 can be given as 2NaOH + CO2 → Na2CO3 + H2O
Question 17.
A sulphate salt of Group % element of the Periodic Table is a white, soft substance, which can be moulded into different shapes by making its dough. When this compound is left in open for some time, it becomes a solid mass and cannot be used for moulding purposes. Identify the sulphate salt and why does it show such a behaviour? Give the reaction involved,
Answer:
The substance which is used for making different shapes is Plaster of Paris. Its chemical name is calcium sulphate – hemihydrate (CaSO4. \(\frac {1}{2}\) H2O). The two formula unit of CaSO4 share one molecule of water. As a result, it is soft. When it is left open for some time, it absorbs moisture from the atmosphere and forms gypsum, which is a hard solid mass.
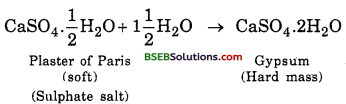
Question 18.
Identify the compound X on the basis of the reactions given below. Also, write the name and chemical formulae of A, B and C.
Answer:
X – NaOH (Sodium hydroxide)
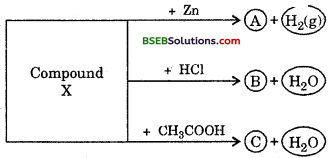
A – Na2ZnO2 (Sodium zincate)
B – NaCl (Sodium chloride)
C – CH3COONa (Sodium acetate)
![]()