Bihar Board Class 12 Chemistry Solutions Chapter 11 Alcohols, Phenols and Ethers Textbook Questions and Answers, Additional Important Questions, Notes.
BSEB Bihar Board Class 12 Chemistry Solutions Chapter 11 Alcohols, Phenols and Ethers
Bihar Board Class 12 Chemistry Alcohols, Phenols and Ethers Intext Questions and Answers
Question 1.
Classify the following as primary, secondary and tertiary alcohols.
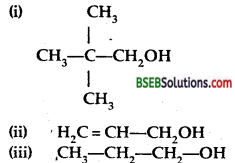

Answer:
(i), (ii) and (iii) are primary alcohols as they have -CH2OH group in them.
(iv) and (v) are secondary alcohols as they have ![]() group in them.
group in them.
(vi) is the tertiery alcohol as they have ![]() group them.
group them.
Question 2.
Identify allylic alcohols in the above examples. :
Answer:
(ii) and (vi) are examples of allylic alcohols.
Question 3.
Name the following compounds according to IUPAC system.

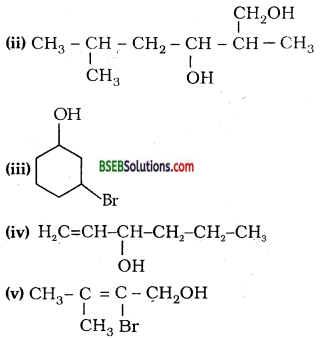
Answer:
(i) 3-Chloromethyl-2-isopropylpentan-l-ol.
(ii) 2,5-Dimethylhexane-l, 3-diol,
(iii) 3-Bromocyclohexanol, (iv) Hex-1-en-3-ol
(v) 2-Bromo-3-methylbut-2-en-l-ol.
Question 4.
Show how are the following alcohols prepared by the reaction of a suitable Grignard reagent on methanol.

Answer:
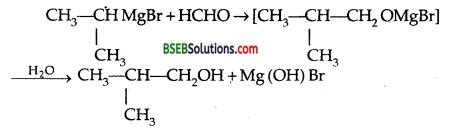

Question 5.
Write structures of the products of the following reactions,
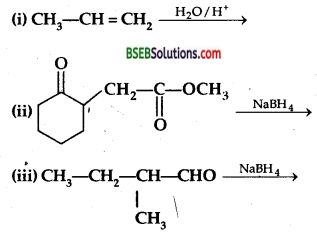
Answer:
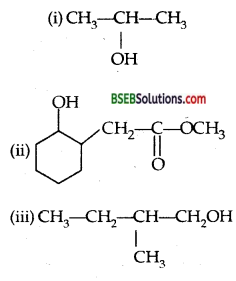
![]()
Question 6.
Give structures of the products you would expect when each of the following alcohol reacts with
(a) HCl-ZnCl2
(b) HBr,
(c) SOCl2.
(i) Butan-l-ol,
(ii) 2-Methyl butan-2-ol
Answer:
(i) Butan-l-ol with ZnCl2-HCl:
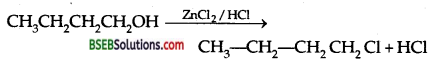
Butan-l-ol with HBr:
CH3 CH2 CH2 CH2 OH + HBr → CH3CH2 CH2 CH2 Br + H2O Butan-l-ol with SOCl2
CH3CH2CH2CH2OH+SOCl2 → CH3CH2CH2CH2Cl+SO2+HCl
(ii) 2-Methyl butan-2-ol
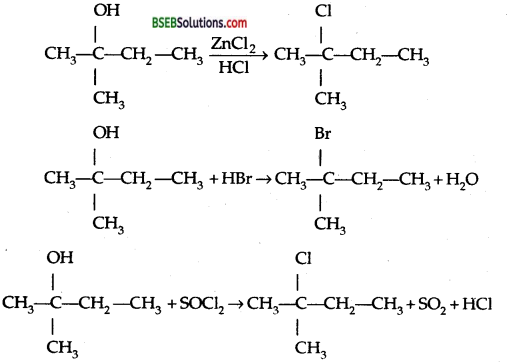
Question 7.
Predict the major product of acid catalysed dehydration of (i) 1-Methyl cyclohexanol, and (ii) butan-l-ol.
Answer:

(ii) Butan-l-ol gives but-l-ene as the major products.

Question 8.
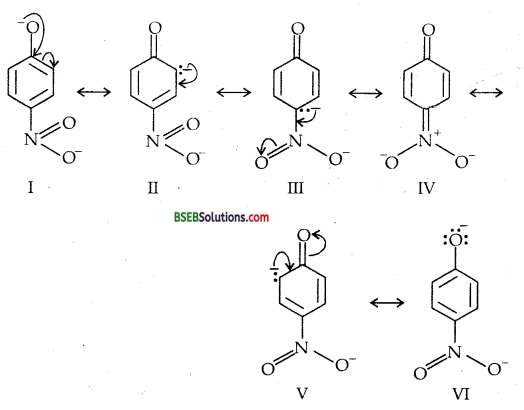
Ortho and Para nitro-phenols are more acidic than phenol.Draw the resonance structures of the corresponding phenoxide ions.
Answer:
(a) Resonance structures of phenoxide ion:
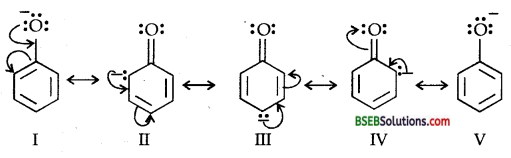
(b) Resonance structures of ortho-nitro phenoxide ion
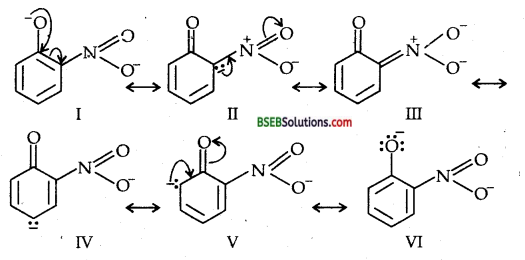
(c) Resonance structures of para-nitro phenoxide ion.
Question 9.
Write the equations involved in the following reactions: (i) Reimer Tiemann reaction, (ii) Kolbe’s reaction
Answer:
(i) Reimer-Tiemann reaction
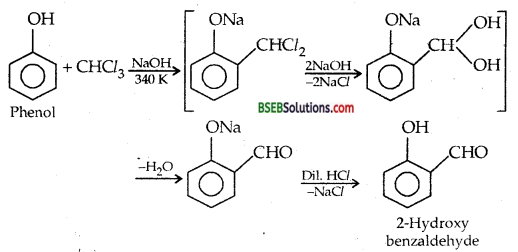
(ii) Kolbe’s Reaction-
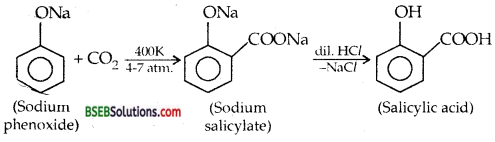
Question 10.
Write the reaction of Williamson synthesis of 2-ethoxy- 3-methyl pentane starting from ethanol and 3-methyl pentan-2-oI.
Answer:
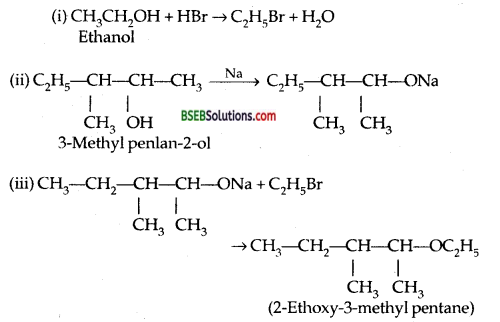
Question 11.
Which of the following is an appropriate set of reactants for the preparation of l-methoxy-4-nitrobenzene and why?
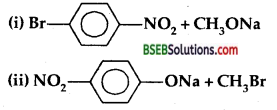
Answer:
(ii) is an appropriate set of reactants to prepare l-methoxy-4- nitrobenzene. It is due to the fact that aryl halides are much less reactive towards nucleophilic substitution reactions than alkyl halides.
Question 12.
Predict the products of the following reactions:
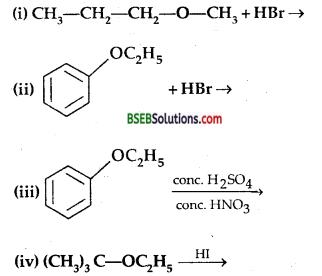
Answer:
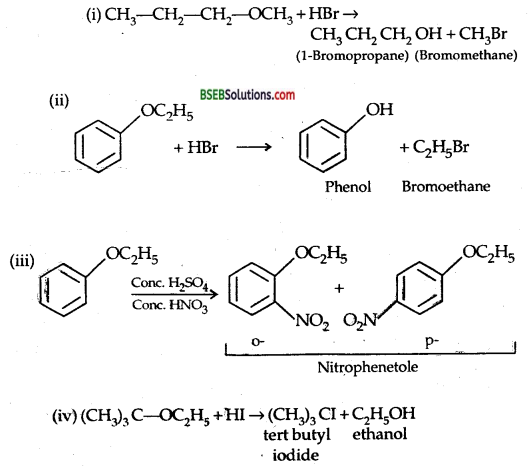
Bihar Board Class 12 Chemistry Alcohols, Phenols and Ethers Text Book Questions and Answers
Question 1.
Write IUPAC names of the following compounds:
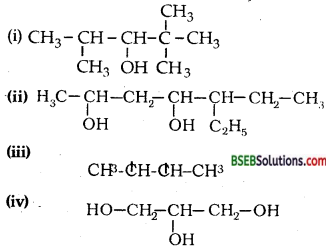

Answer:
(i) 2,2,4-Trimethyl pentaii-3-ol,
(ii) 5- Ethylheptane-2,4-diol,
(iii) Butane-2, 3-diol,
(iv) Propane-1, 2, 3-triol,
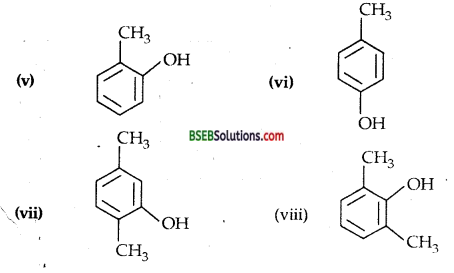
(v) 2-Methyl phenol,
(vi) 4-Methyphenol,
(vii) 2,5-Dimethylphenol,
(viii) 2,6-Dimethylphenol,
(ix) l-Methoxy-2-methyl propane,
(x) Ethoxybenzene,
(xi) 1-Phenoxyheptane,
(xii) 2-Ethoxybutane.
Question 2.
Write structures of the compounds whose IUPAC names are as follows: .
(i) 2-Methylbutan-2-ol
(ii) l-Phenylpropan-2-ol
(iii) 3,5-Dimethylhexane-l, 3,5-triol
(iv) 2,3-Diethyl phenol
(v) 1-Ethoxypropane
(vi) 2-Ethoxy-3-methylpentane
(vii) Cyclohexylmethanol
(viii) 3-Cyclohexylpentan-3-ol
(ix) Cyclopent-3-en-l-ol
(x) 3-Chloromethyl pentan-l-ol.
Answer:
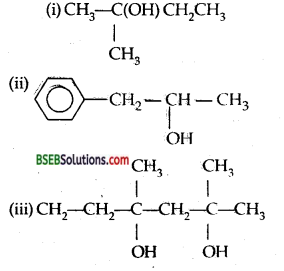
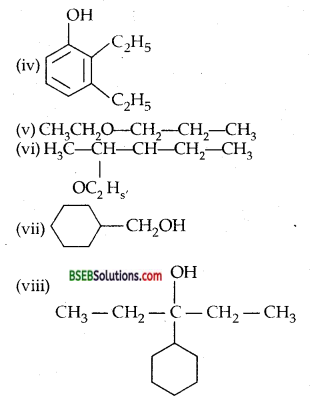
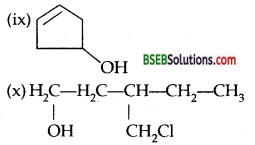
Question 3.
(i) Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
(ii) Classify the isomers of alcohols in question 3(i) as primary, secondary, and tertiary alcohols.
Answer:
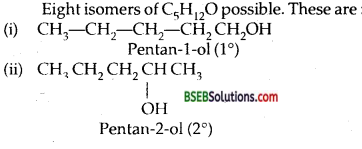
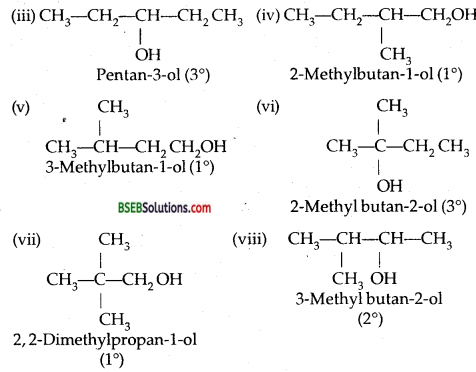
Question 4.
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
Answer:
Though butane and propanol have comparable molecular masses (58 and 60 respectively) yet propanol has higher boiling than butane because propanol shows hydrogen bonding.
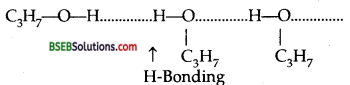
due to which its molecules get associated and boil at higher temperature than butane because it shows no hydrogen bonding.
The molecules of butane are held together by only weak van der Waals forces of attraction.
![]()
Question 5.
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
Answer:
Alcohols can form H-bonds with water and break the H- bonds already existing between water molecules. Therefore they are soluble in water.
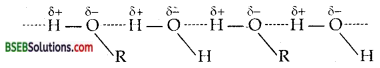
On the other hand, hydrocarbons cannot form H-bonds with water and hence are insoluble in water.
Question 6.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Answer:
Hydroboration-oxidation-Alkenes react with BH3 of diborane (B2H6) to. form trialkyl boranes which on subsequent treatment with alkaline H2O2 gives alcohols. Thus
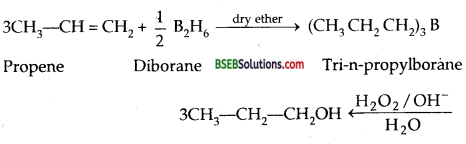
This two-step process is called hydroboration-oxidation and gives alcohols corresponding to ANTl-M ARKOUNIKOV’s addition of water to alkenes. During oxidation of trialkyl borane, boron is replaced by – OH group. The yield of alcohol is excellent and the product can be easily isolated.
Question 7.
Give the structures and IUPAC names of monohydric phenols of molecular formula C7H8O.
Answer:
Monohydric phenols of molecular formula C7H8O are:
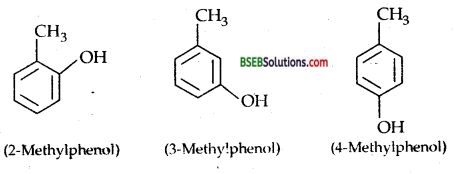
Question 8.
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Answer:
o-Nitrophenol is steam volatile because of intramolecular 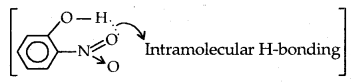 Intramolecular H-bonding Therefore B.Pt. of o-nitrophenol is lower than para nitrophenol which shows intermolecular H-bonding and has higher B.Pt.
Intramolecular H-bonding Therefore B.Pt. of o-nitrophenol is lower than para nitrophenol which shows intermolecular H-bonding and has higher B.Pt.
Question 9.
Give the equations of reactions for the preparation of phenol from cumene.
Answer:
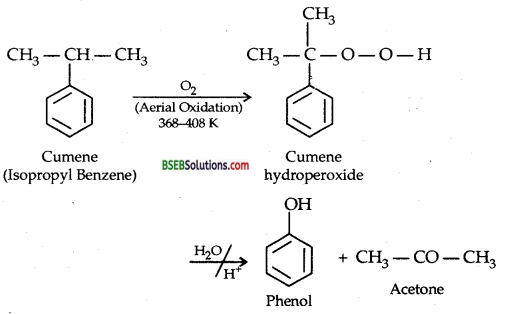
Question 10.
Write chemical reaction for the preparation of phenol from Chlorobenzene.
Answer:
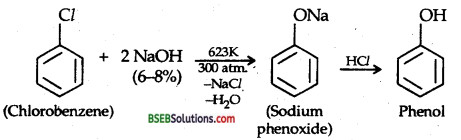
Question 11.
Write the mechanism of hydration of ethene to yield ethanol.
Answer:
Ethene undergoes hydration in the presence of H2SO4 to yield ethyl alcohol.
![]()
Mechanism: Step I: Protonation of alkene to form carbocation by electrophilic attach of H3O+ ion.
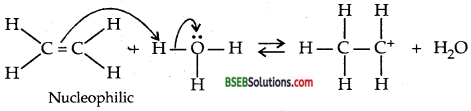
Step II. Nucleophic attack of water on carbocation

Step III. Deprotonation to form an alcohol
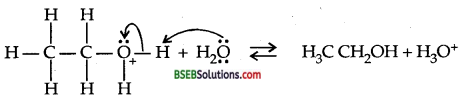
![]()
Question 12.
You are given benzene, cone. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
Answer:
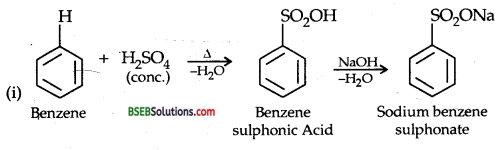
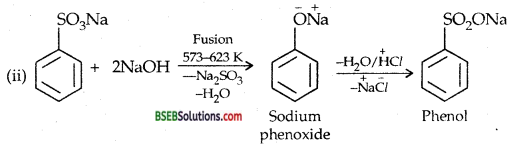
Question 13.
Show how will you synthesise (i) 1-Phenyl ethanol from a suitable alkene, (ii) Cyclohexylmethanol using an alkyl halide by an SN2 reaction.
(iii) Pentan-l-ol using a suitable alkyl halide?
Answer:
(i) 1-Phenyl ethanol from a suitable alkene: 1-Phenylethene is subjected to hydration
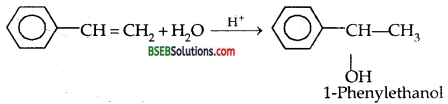
(ii) Cyclohexylmethanol by SN2


Question 14.
Give two reactions that show the acidic nature of phenol. Compare acidity of phenol with that of ethanol.
Answer:
Acidic nature of phenol is shown by the following reactions :
(a) (i) Reactions with active metals like Na, K, Mg etc:
Hydrogen gas is liberated

(ii) Reaction with aqueous NaOH: Phenol behaves like a weak add neutralizing NaOH-an alkali and forming salt.
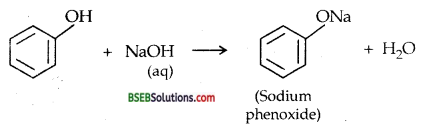
(b) Comparison of acidity of phenol with that of ethanol
C2H5OH + NaOH → No reaction
The above reaction of phenol with aqueous NaOH indicates that phenol is a stronger acid than ethanol.
The ionisation of phenol and ethanol is as follows :
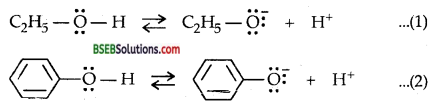
Whereas phenol and phenoxide ion in (2) both show resonance. The resonance stabilization of phenoxide is more than that of phenol thus shifting equilibrium of equation (2) towards right giving H+ ions.
On the other hand, neigther ethoxide ion nor ethanol show any resonance, i.e., negative charge in ethoxide ion is localised on oxygen, it is delocalised in phenoxide ion. The delocalisation of negative charge makes phenoxide ion more stable than ethoxide ion.
Moreover, Pka of ethanol is 15.9 whereas that of phenol is 10.0. It suggests that phenol is million times more acidic than ethanol.
Question 15.
Explain why is ortho nitrophenol more acidic than ortho methoxy phenol?
Answer:
Due to strong – R and -I-effect of the nitro group (- NO2) electron density in the O-H bond decreases and hence the loss of a proton becomes easy.
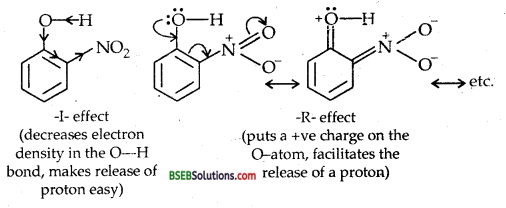
Further, after the loss of a proton, the o-nitro phenoxide ion left behind is stabilized by resonance.
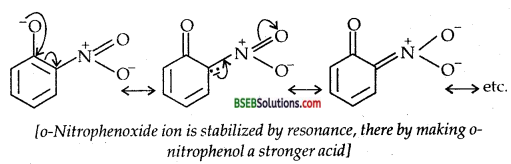
In contrast, due to + R effect of the -OCH3 group, it increases the electron density in the O-H bond thereby making the loss of proton difficult.
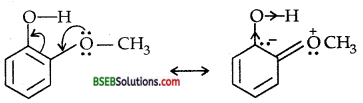
Furthermore, the o-methoxy phenoxide ion left after the loss of a proton is destabilized by resonance.
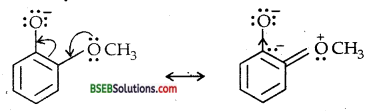
The two negative charges repel each other thereby destabilizing the-methoxy phenoxide ion. Thus o-nitrophenol is more acidic than o- methoxy phenol.
![]()
Question 16.
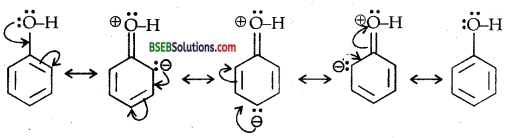
Explain how does the -OH group attached to d carbon of benzene ring activate it towards electrophilic substitution?
Answer:
The -OH group attached to the benzene ring in phenol activates it towards electrophilic substitution. It also directs the incoming group to ortho and para positions in the ring as these positions become electron-rich due to the electronic effect and resonance effect caused by -OH group.
Question 17.
Give equations of the following reactions:
(i) Oxidation of propan-l-ol with alkaline KMnO4 solution.
(ii) Bromine in CS2 with phenol.
(iii) Dilute HNO3 with phenol.
(iv) Treating phenol with chloroform in presence of aqueous NaOH.
Answer:

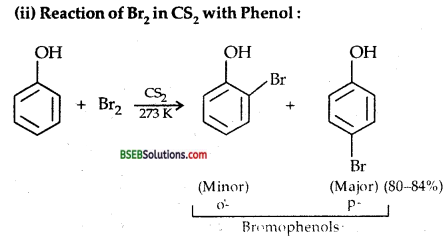
(iii) Action of dilute HNO3 on Phenob-With dil. HNO3 at low temp. (298 K) phenol yields a mixture of ortho (40%) and para (15%) nitrophenols.
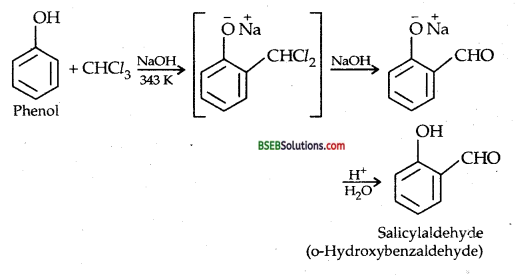
(iv) Treating phenol with CHCl3 and aqueous NaOH at 343 K: REIMER-TIEMANN REACTION
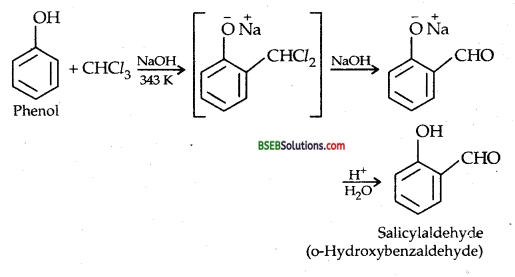
A small amount of p-hydroxybenzaldehyde is also obtained.
Question 18.
Explain the following with an example:
(i) Kolbe’s reaction
(ii) Reimer-Tiemann reaction
(iii) Williamson ether synthesis
(iv) Unsymmetrical ether.
Answer:
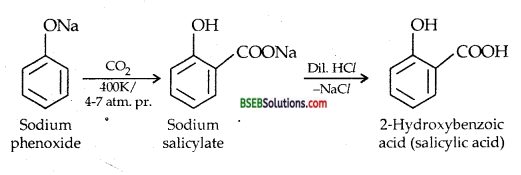
(i) KOLBE’s Reaction-Sodium phenoxide when heated with CO2 at 400 K under a pressure of 4-7 atmospheres followed by acidification gives 2-hydroxy benzoic acid (salicylic acid) as the main product along with a small amount of 4-hydroxybenzoic acid. This reaction is called KOLBE REACTION.
(ii) REIMER-TIEMANN REACTION-Treatment of Phenol with chloroform in presence of aqueous sodium (or potassium) hydroxide at 340 K followed by hydrolysis of the resulting product gives 2-hydroxy benzaldehyde (salicylaldehyde) as the major products.
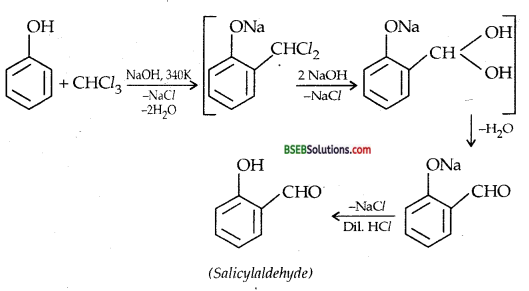
B. CCl4: If CCl4 is used in place of CHCl3, salicylic acid is the main product.
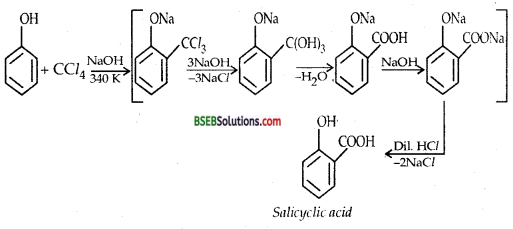
A small amount of p-hydroxybenzoic acid is also obtained. MECHANISM Reimer-Tiemann is an electrophilic substitution reaction and occurs through the following steps :
(i) Generation of electrophile:
HO–+CHCl3 ⇌ H2O + CCl3–→: CCl2 + Cl– Dichlorocarbene (electrophile)
(ii) ELECTROPHILIC SUBSTITUTION-
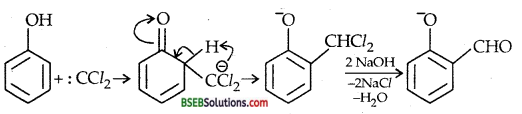
(iii) Williamson ether synthesis—It is an important laboratory method for the preparation of symmetrical and unsymmetrical ethers.
R-X + R’ONa → R-O-R’ + NaX.
It is involves SN2 attack of an alkoxide ion on primary alkyl halide.
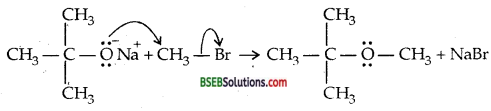
(iv) Unsymmetrical ether is one in which two Rs or two aryl groups attached to ether linkage -O- are different.
For example- R-O-R’ R ≠ R’
Ar-O-R

Question 19.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Answer:
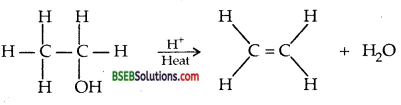
Mechanism: (i) Formation of protonated alcohol
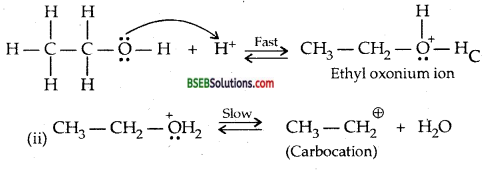
Formation of carbocation being the slow step is the rate-determining
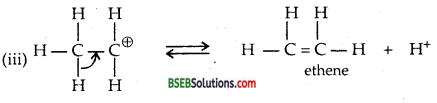
![]()
Question 20.
How are the following conversions carried out?
(i) Propene → Propan-2-ol
(ii) Benzyl chloride → Benzyl alcohol
(iii) Ethyl magnesium chloride → Propan-l-ol
(iv) Methyl magnesium bromide →2-methyl propane-2-ol
Answer:

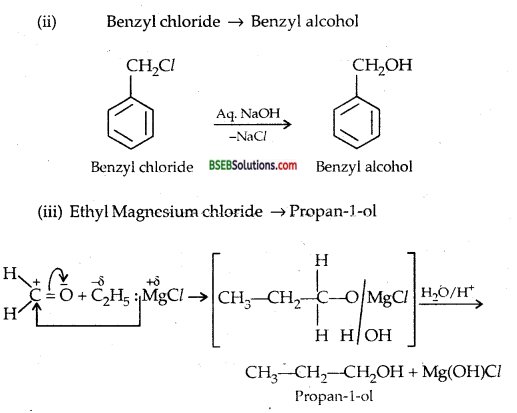
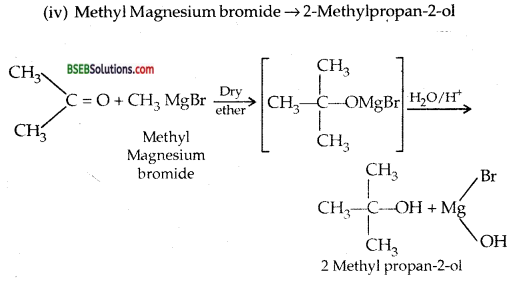
Question 21.
Name the reagents used in the following reactions:
(i) Oxidation of a primary alcohol to carboxylic acid
(ii) Oxidation of a primary alcohol to aldehyde
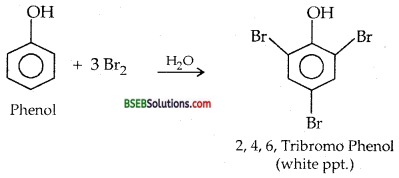
(iii) Bromination of phenol to 2,4,6-tribromophenol
(iv) Benzyl alcohol to benzoic acid
(v) Dehydration of propan-2-ol to propene
(vi) Butan-2-one to butan-2-ol
Answer:
(i) Acidified potassium dichromate or neutral, acidic or alkaline potassium permanganate (KMnO4).
(ii) Pyridinium chlorochromate (PCC).
(iii) Aqueous Bromine
(iv) Acidified or alkaline KMnO4.
(v) Cone. H2SO4 at 433-443 K.
![]()
Question 22.
Give reasons for the higher boiling point of ethanol in comparison to methoxymethane.
Answer:
Ethanol molecules get associated due to H-bonding
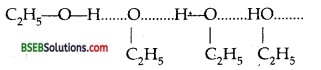
whereas hydrogen bonding is not present in methoxy methane.
Question 23.
Give IUPAC names of the following ethers:
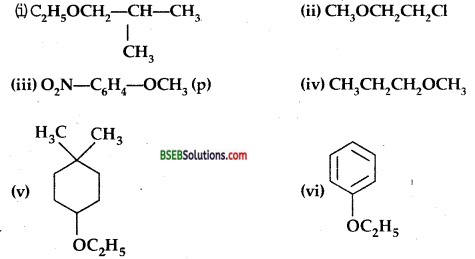
Answer:
(i) l-Ethoxy-2-methyl-propane
(ii) 2-Chloro-l-methoxy ethane
(iii) 4-Nitroanisole.
(iv) 1-Methoxypropane
(v) l-Ethoxy-4,4-dimethyl cyclohexane
(vi) Ethoxybenzene.
Question 24.
Write the names of reagents and equations for the preparation of the following ethers by Williamson’s synthesis:
(i) 1-Propoxypropane
(ii) Ethoxybenzene
(iii) 2-Methoxy-2 methylpropane
(iv) 1-Methoxyethane
Answer:
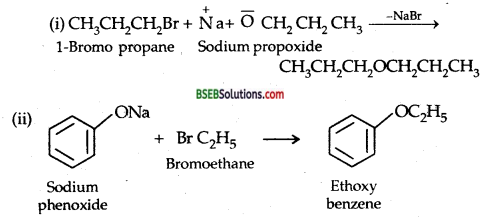
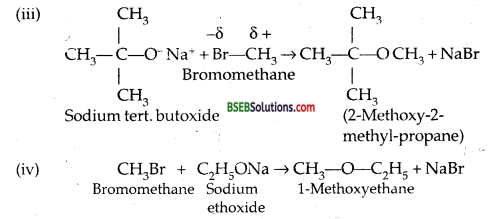
Question 25.
Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
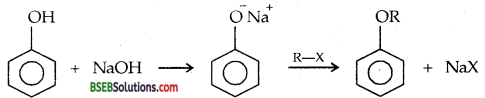
Ans. Williamson Synthesis is based on the nucleophilic substitution of alkyl halides in which halogen is replaced by alkoxy group. It involves the reaction of RX with sodium or pot. salt of alcohol or phenol.
Na+ OR’→ R-O-R’ + NaX
Alkyl halide Sodium ethoxide
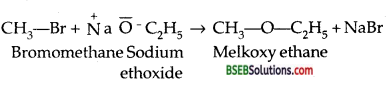
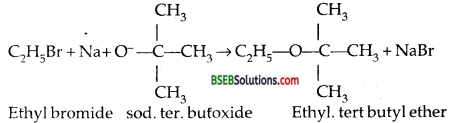
It may be noted that for preparing unsymmetrical ethers, a proper choice of the reactants is quite necessary. The halide used in this reaction should preferably be primary because secondary and tertiery halides readily undergo elimination reaction in the presence of a strong base to form alkenes as the major product. For example, if we have to prepare ethyl tertiery butyl ether, then we shall use ethyl bromide and sodium tertiery butoxide.
If we use tert. butyl chloride and sodium ethoxide as reactants, the major products would be isobutylene and ethanol.
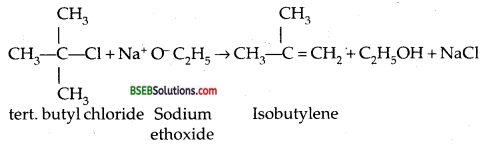
It is because alkoxides are NOT ONLY NUCLEOPHILES but also strong bases. They react with tertiery alkyl halides leading to elimination reaction.
Similarly, aryl halides and sodium alkoxide CANNOT be used for preparing phenolic ethers because of very less reactivity of aryl halides towards nucleophilic substitution.
Question 26.
How is 1-pro poxypropane synthesized from propane-1 – ol? Write mechanism of this reaction.
Answer:
1- Propoxy propane can be prepared from propan-l-ol by Williamson synthesis
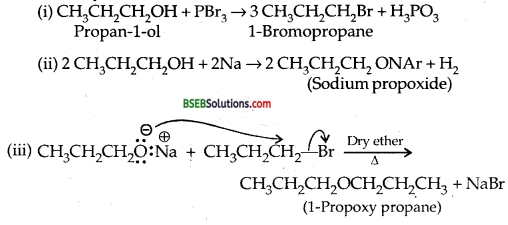
Question 27.
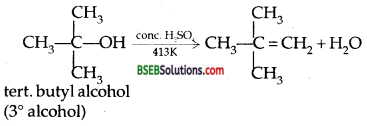
Preparation of ethers by acid dehydration of secondary c r tertiary alcohols is not a suitable method. Give reason.
Answer:
Ethers cannot be conveniently prepared from the acidic dehydration of secondary alcohols as they are formed only in low yields. For example secondary alcohols on treatment with cone. H2S04 at413lK mainly gives alkenes with low yields of ether.

Thus the order of acid dehydration of alcohols to give ethers follows
the sequence primary > secondary> tertiery.
This method of acid dehydration is also useless to prepare unsymmetrical ethers of the type RORt as complex mixtures are obtained:
![]()
This method of preparing ether is suited to industries as reaction conditions can be controlled and is of little utility in the laboratory.
Question 28.
Write the equation of the reaction of hydrogen iodide with
(i) 1-pro poxypropane,
(ii) methoxybenzene and
(iii) benzyl ethyl ether.
Answer:
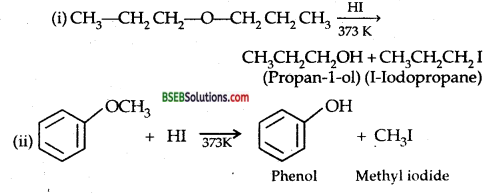
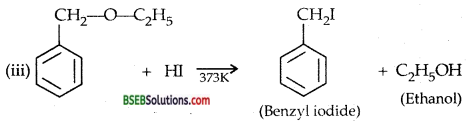
![]()
Question 29.
Explain the fact that in aryl alkyl ether (i) the alkoxy group activates the benzene ring towards electrophilic substitution and (ii) it directs the incoming substituents to ortho and para positions in benzene ring.
Answer:
In aryl alkyl ethers, the + R effect of the alkoxy group (OR) increases the electron density in the benzene ring (as shown below) thereby activating the benzene ring towards electrophilic substitution reactions.
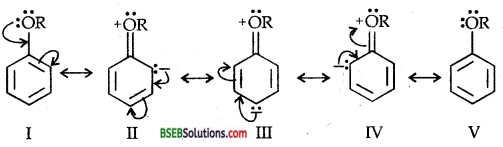
Since the electron density increases more at the two ortho and one para positions as compared to m-positions, therefore, electrophilic substitution reactions mainly occur at o- and p-positive.
Question 30.
Write mechanism of the reaction of HI with methoxymethane.
Answer:
CH3-O-CH3 + HI→CH3I + CH3OH
Step I: Ethers being Lewis bases, undergo protonation to form oxonium ion

Step II:
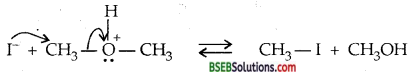
When HI is used in excess and at high-temperature methanol reacts with another molecule of HI and is converted to methyl iodide. Step III:
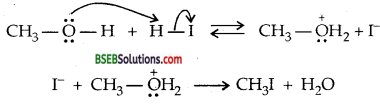
Question 31.
Write equations of the following reactions:
(i) Friedel Crafts reaction-alkylation of anisole
(ii) Nitration of anisole
(iii) Bromination of anisole in ethanoic acid medium,
(iv) Friedel-Craft’s acetylation of anisole.
Answer:
Friedel-Cratts reaction-alkylation of anisole
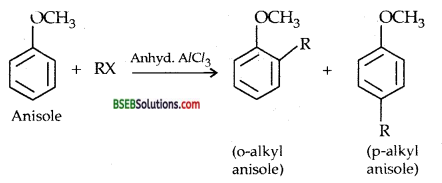
(ii) Nitration of anisole-Anisole reacts with a mixture of cone. H2SO4 and cone. HNO3 to yield a mixture of ortho and para nitro-anisole.
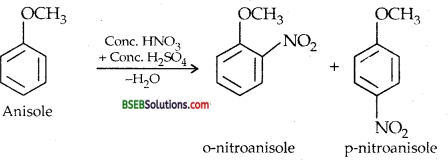
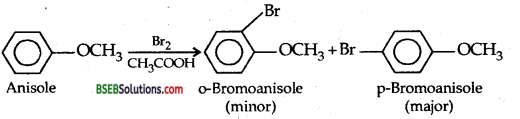
(iii) Bromination of anisole in ethanoic acid medium-Phenyl alkyl ethers undergo usual halogenation in the benzene ring, e.g., anisole undergoes bromination with bromine in ethanoic acid even in the absence of Iron (III) bromide catalyst. It is due to the activation of benzene ring by the methoxy group. Para isomer is obtained in 90% yield.
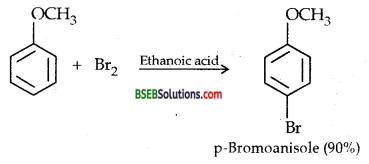
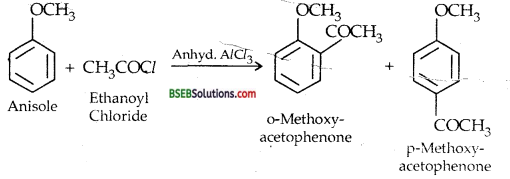
(iv) Friedel crafts acetylation of Anisole-Anisole undergoes the usual electrophilic substitution reaction when acetyl chloride reacts with it in the presence of anhydrous AlCl3 which acts as a LEWIS ACID. The acetyl group is dlfected.to both ortho and pr -a positions in the benzene ring.
Question 32.
Show how would you synthesize the following alcohols from appropriate alkenes?

Answer:
(i) From 1-Methyl cyclohexene
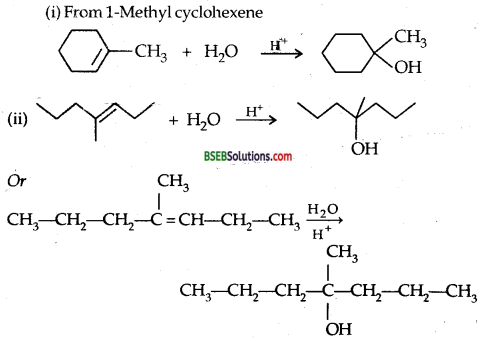
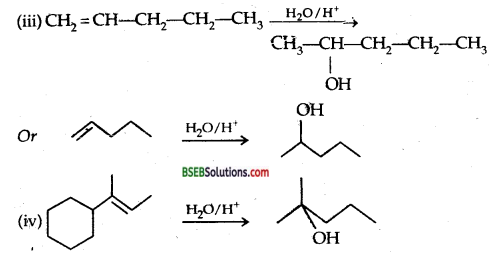
Question 33.
When 3-methyl butane-2-ol is treated with HBr following reaction takes place;
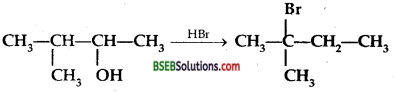
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a move stable tertiery carbocation by a hydride ion shift from 3rd carbon atom.)
Answer:

Bihar Board Class 12 Chemistry
Aldehydes, Ketones and Carboxylic Acids
Additional Important Questions and Answers
Very Short Answer Type Questions
Question 1.
How much bromipe (in moles) is needed to produce 2,4,6- tribromophenol from one mole of phenol?
Answer:
3 Moles.
Question 2.
What is the order of dehydration of primary, secondary and tertiary alcohols.
Answer:
3° > 2° > 1° i.e., tert. alcohol > secondary alcohol > primary alcohol.
Question 3.
What is the IUPAC name of HC ≡ C-CH2OH.
Answer:
Prop-2-yn- l-ol.
Question 4.
Mention two important uses of methanol. (A.I.S.B. 2002)
Answer:
- For denaturing ethyl alcohol,
- in the manufactures of formaldehyde (HCHO) which is further widely used to prepare bakelite, resins etc.
Question 5.
How will you convert ethanol into ethylene. (P.S.B. 2002)
Answer:
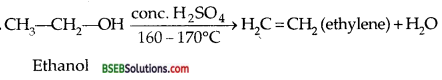
Question 6.
Anhydrous CaCl2 is not recommended as a drying agent for alcohols and amines. M. L. N. R. 1991)
Answer:
Alcohols and amine combine with anhydrous CaCl2 to form complexes. For example with C2H5OH, CaCl2 gives a complex CaCl2. 3C2H5OH.
![]()
Question 7.
Why ethers are insoluble in water? (P.S.B. 97)
Answer:
Ethers are insoluble in water because of bigger size of the alkyl groups, the oxygen atom in ethers fail to form intermolecular hydrogen bonds with water.
Question 8.
Of the two hydroxy organic compounds ROH and R’OH the first is basic and the other is acidic in behaviour. How is R difference from R’? (P.S.B. 2000)
Answer:
When R = alkyl; ROH behave as a LEWIS BASE.
When R’ = aryl; R’OH behaves a Bronsted acid.
Question 9.
Phenol has smaller dipole moment than methanol. Explain. (D.S.B. 2002)
Answer:
Due to electron-withdrawing effect of the benzene ring the C-O bond in Phenol is less polar but in case of methanol, due to electron-releasing effect of CH3 group C-O bond is more polar.
Question 10.
Why is common phenol more acidic than ethyl alcohol? (A.I.S. B. 1993)
Answer:
Phenoxide ion of phenol is RESONANCE STABILISED whereas alkoxide ion is not.
Question 11.
Name the alkyl halide and sodium alkoxide used to synthesise tert butyl ethyl ether. (H.S.B. 93, C.B.S.E. Sample Paper 1997)
Answer:
Ethyl bromide and sodium tert. butoxide.
Question 12.
Diethyl ether does not react with sodium. Explain x (B.I.T. Ranchi 1992)
Answer:
Since diethyl ether does not contain an active hydrogen attached to oxygen like in alcohols and phenols which can be displaced as H2.
Question 13.
Give the structural formula of 2-ethoxypropane.(H.S.B. 94)
Answer:
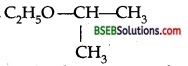
Question 14.
Write the structure of perchlorodiethyl ether. (H.S.B. 97)
Answer:
CCl3 CCl2 OCCl2 CCl3.
Question 15.
Name the reagent used to convert bromoethane to ethoxyethane. (P.S.B. 2002)
Answer:
Sodium ethoxide (CH3CH2O– Na+).
Question 16.
How many sigma bonds are present in 3-methyl phenol?
Answer:
Sixteen.
Short Answer Type Questions
Question 1.
Give the IUPAC names of
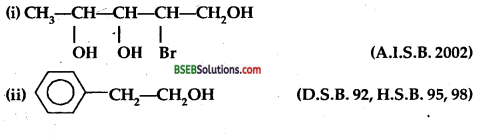
Answer:
(i) 2-Bromopentane-l, 3,4 triol.
(ii) 2-phenyl ethanol.
Question 2.
Give the I.U.P.A.C. names of

Answer:
(i) 2-Hydroxy toluene or 2-Methyl phenol
(ii) 2,3-Dimethyl butane-2,3-diol.
Question 3.
Give the structures of:
(i) Isobutyl alcohol
(ii) Ethoxy propane.
Answer:
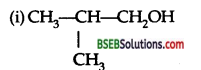
Question 4.
How will you prepare 2-propanone from 2-propanol?
Answer:

Question 5.
How will you prepare 2-propanol from 2-propanone.
Answer:
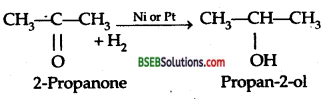
Question 6.
Arrange the following in increasing order of basicity? (I.I.T.92)
H2O, OH–, CH3OH, CH3O–.
Answer:
H2O, CH3OH, OH–,CH3O–.
![]()
Question 7.
Give the structure of the major organic product when 3- ethyl pent-2-ene is reacted with Hg (OAC)2/H2O, NaBH4. (I.I.T. 96)
Answer:
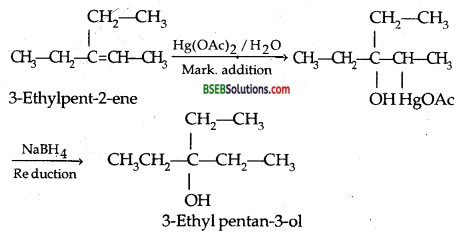
Question 8.
How will you convert acetylene to ethanol?
Answer:
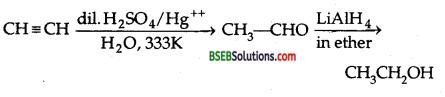
Question 9.
How will you convert 1-propanol to 2-bromopropane?
Answer:
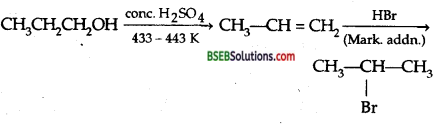
Question 10.
What happens when diethyl ether is treated with dil. H2SO4 and is treated under pressure.
Answer:

Question 11.
Write the chemical equation when benzyl phenyl ether reacts with HI at 375 K.(P.S.B. 2001)
Answer:
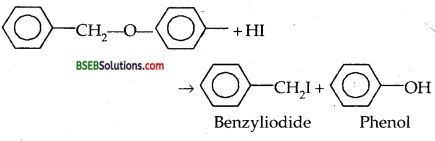
Question 12.
Give the I.U.P. A.C. name of CH3O. CH2CH2. OC2H5.
Answer:
2-Ethoxy-l-methoxy ethane.
Question 13.
Convert phenol to acetophenone. (D.S.B. 2004 S, 2005 S)
Answer:
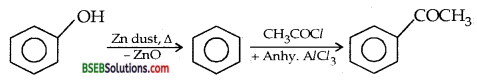
Question 14.
Convert phenol to toluene (D.S.B. 2005 S)
Answer:
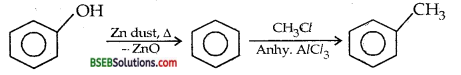
Question 15.
Convert benzene to anisole
Answer:
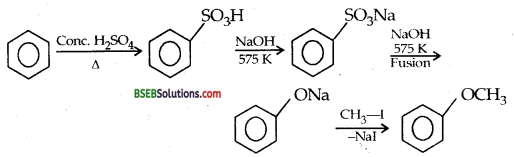
![]()
Question 16.
How. will you distinguish ethanol (C2H5OH) and phenol (C6H5-OH) ? (AISB1985, DSB 87, PSB 89, HPSB 2004, H.S.B. 2005)
Answer:
(i) Litmus test-Phenol being acidic turns blue litmus red while ethanol being neutral does not give this test.
(ii) FeCl3 test-Phenol gives a violet colouration with neutral FeCl3 solution while ethanol does not.
(iii) Br2-water Test-Phenol decolourised bromine water giving white ppt. of 2,4,6-Tribromophenol but ethanol does not

Question 17.
How will you distinguish between methanol and ethanol? (IIT 85, AISB 86, 95, DSB 88, 89, 2001, HPSB 88 CBSE Sample Paper 97, P.S.B. 89,91,2001).
Answer:
Ethanol responds to iodoform test as given below, where as methanol does not.
CH3CH2OH + NaOI → CH3CHO + Nal + H2O
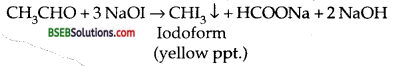
Questions Based Upon Structures
Question 1.
Compound (A) C4H10O is found to be soluble in sulphuric acid. (A) does not react with sodium metal or potassium permanganate. When (A) is heated with excess of HI it is converted into a single alkyl halide. What is the structural formula of A?
Answer:
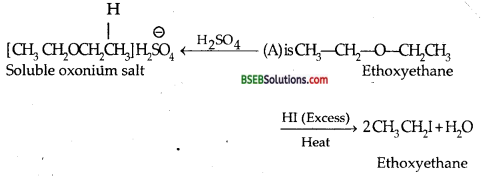
Question 2.
Give the structures of all ethers having the molecular formula C4H10O and their I.U.P.A.C. names.
Answer:
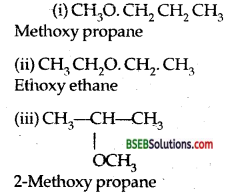
Question 3.
An organic compound A (C2H6O) reacts with sodium to form a compound B with the evolution of H2 and gives a yellow compound C when heated with I2 and NaOH. When heated with cone. H2SO4 at 413 K, if gives a compound D (C4H10C) which on treatment with HI at 373 K gives E, D is also obtained when B is heated with E. Identify A, B, C, D, E and write equations for the reactions involved.
Answer:
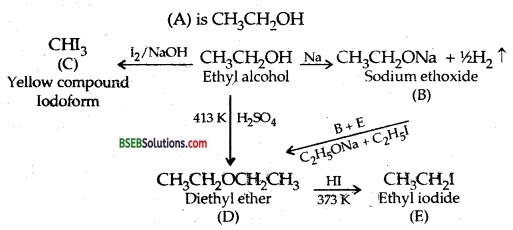
![]()
Question 4.
How will you synthesis 4-methoxyphenyl from bromobenzene in not more than 5 steps. State clearly the reagents used in each step and show the structures of intermediate compounds in your synthetic scheme. (I.I.T. 2001)
Answer:
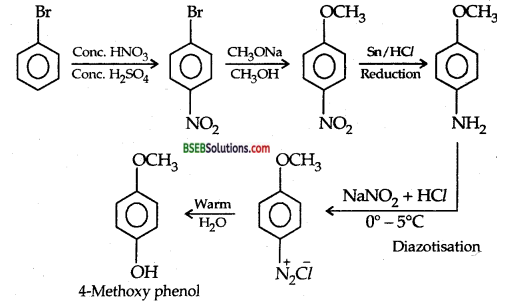
Question 5.
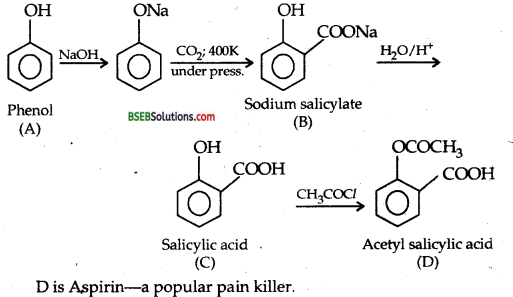
An organic compound ‘A’ having the molecular formula C6H6O gives a characteristic colour with aqueous FeCl3 solution. When A is treated with CO2 and NaOH at 400 K under pressure ‘B’ is obtained. The compound B on acidification gives compound ‘C’ which reacts with acetyl chloride to form ‘D’ which is a popular pain killer. Deduce the structures of A, B, C and O.
Answer:
Since the compound ‘A’ C6H6O gives a characteristic colour with aqueous FeCl3 solution, it is a phenol C6H5-OH. The sequence of reactions are:
Question 6.
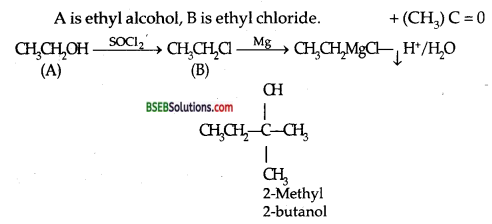
A compound (A) reacts with thionyl chloride to give compound B. (B) reacts wth Mg with Grignard reagent which is treated with acetone and the product is hydrolysed to give 2-methyl-2-butanol. What are (A) and (B) compounds?
Answer:
Long Answer Type Questions
Question 1.
(a) How many types of isomers are shown by alcohols? Explain giving example.
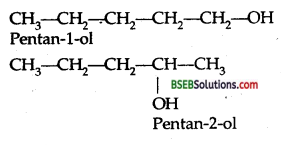
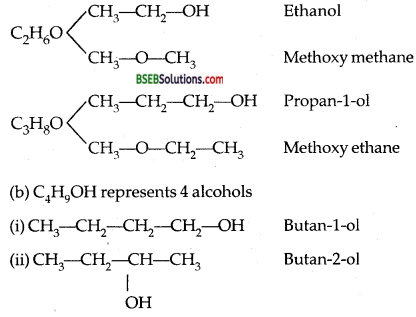
(b) Write down all isomers of alcohols (C4H9OH). Which out of them will show optical isomerism?
Answer:
(a) Alcohols show the following types of isomerisms:
I. Chain isomerism: Alcohols containing 4 or more C atoms exhibit chain isomerism due to the difference in the length of the chain of C atoms.

II. Position isomerism: This isomerism arises due to the difference in the position of the functional group.
III. Functional isomerism: Alcohols show functional isomerism with ethers
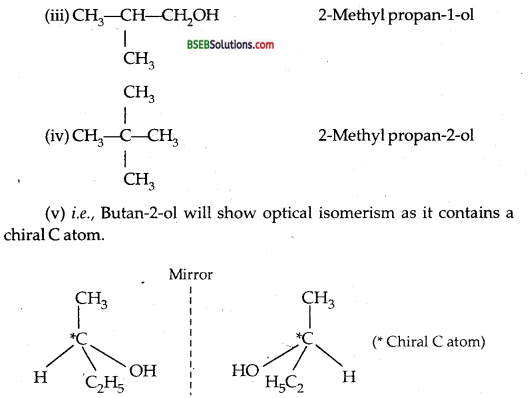
![]()
Question 2.
(i) How will you prepare phenol form
(a) chlorobenzene,
(b) benzene diazonium chloride,
(c) cumene,
(ii) How does it react with
(a) Benzene Diazonium chloride
(b) Ammonia
(c) Benzyl chloride,
(d) Aqueous Bromine.
Answer:
(i) Preparation of Phenol from chlorobenzene: DOW’s PROCESS:
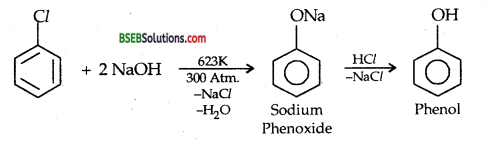
(b) Preparation of Phenol from Benzene Diazonium chloride:
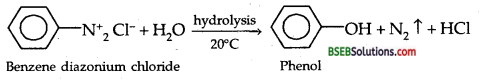
(c) Preparation of Phenol from Cumene

(ii) (a) Reaction of Phenol with Benzene diazonium chloride COUPLING REACTION:
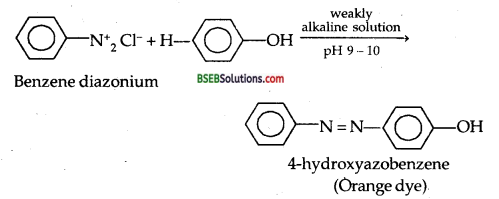
(b) Reaction of Phenol with Ammonia:
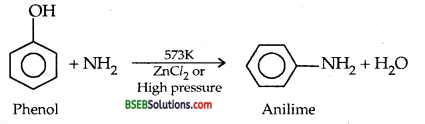
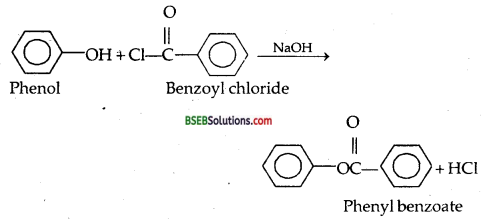
(c) Reaction of Phenol with Benzoyl chloride SCHOTTEN BAUMANN REACTION.
(d) Reaction of Phenol with Aqueous Bromine water-It forms 2, 4,6 Tribromophenol (white ppt).