Bihar Board Class 12 Chemistry Solutions Chapter 14 Biomolecules Textbook Questions and Answers, Additional Important Questions, Notes.
BSEB Bihar Board Class 12 Chemistry Solutions Chapter 14 Biomolecules
Bihar Board Class 12 Chemistry Biomolecules Intext Questions and Answers
Question 1.
Glucose or Sucrose are soluble in water, but cyclohexane or benzene (simple six-membered ring compounds) are insoluble in water. Explain.
Answer:
Glucose or Sucrose contain several hydroxyl groups in their molecules which form Hydrogen bonding with water molecules due to which they dissolve in water. On the other hand compounds like benzene or cyclohexane cannot form hydrogen bonds with water molecules, so they are insoluble in water.
Question 2.
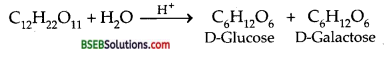
What products are expected when lactose is hydrolysed?
Answer:
Lactose (C12H22O11) on hydrolysis with dilute acid yields an equimolar mixture of D-glucose and D-galactose.
Question 3.
How do you explain the absence of aldehyde group in the pentaacetate of D-glucose?
Answer:
Glucose pentaacetate exists in two aromeric forms as explained below: –
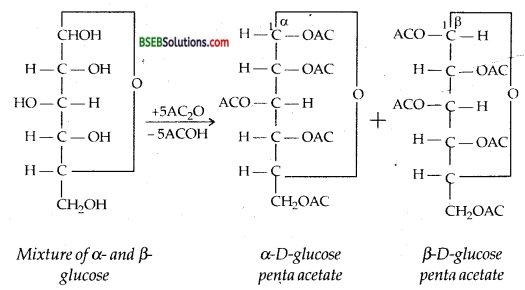
These penta acetates da not have a free – OH group at C1 and hence are not hydrolysed in aqueous solution to produce the open-chain aldehyde form.
Question 4.
The melting points and solubility in water of amino acids are generally higher than that of the corresponding halo acids. Explain.
Answer:
Because of the Zwitter ion structures [internal salt]
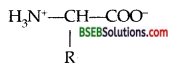
α-amino acids are highly polar and so are high melting crystalline solids. Except at Isoelectric point where the amino acid has least solubility in water, these amino acids are moderately soluble in water again due to their polar character. On the other hand, haloacids being covalent compounds do not form such an ion and so are soluble to a less extent and their melting points are low.
![]()
Question 5.
Where does the water present in the egg go after boiling the egg.
Answer:
The boiling of an egg is a common example of denatination of proteins present in the white portion of an egg.
The albumin present in the white of an egg gets coagulated when the egg is boiled hard. The soluble globular protein present in it is denatured resulting in the formation of insoluble fibrous protein.
Question 6.
Why Vitamin C cannot be stored in our body?
Answer:
Vitamin C is a water-soluble vitamin. Water-soluble vitamins when supplied regularly in the diet cannot be stored in our body because they are readily exerted in urine.
Question 7.
What products would be formed when a nucleotide from DNA containing thymine is hydrolysed?
Answer:
When a nucleotide from DNA containing thymine is completely hydrolysed,the products obtained are:
(i) 2-deoxy-D (-) ribose.
(ii) two pyrimidines i.e., guanine (G) and adenine (A).
(iii) two purines, i.e., thymine (T) and Cytosine (C) and (iv) phosphoric acid.
Question 8.
When RNA is hydrolysed, there is no relationship among the quantities of different bases obtained. What does this fact suggest about the structure of RNA?
Answer:
A DNA molecule has two strands in which the four complementary bases pair each other, i.e., Cytosine (C) always pairs with guanine (G) while thymine (T) always pairs with adenine (A). Therefore when a DNA molecule is hydrolysed, the molar amounts of cytosine is always equal to that of guanine and that of adenine is always equal to that of thymine. Since in RNA there is no relationship between the quantities of four bases (C, G, A and U) obtained, therefore the base pairing principle, i.e., A pairs with U and C pairs with G is not followed. Therefore unlike DNA, RNA has a single strand.
Bihar Board Class 12 Chemistry Biomolecules Text Book Questions and Answers
Question 1.
What are monosaccharides?
Answer:
A carbohydrate that cannot be hydrolysed further to give simpler unit of polyhydroxy aldehyde or ketone is called a monosaccharide. Common examples are glucose, fructose, ribose etc.
Question 2.
What are reducing sugars?
Answer:
Reducing sugars are those which reduce Fehling’s solution and Tollen’s reagent. All monosaccharides whether aldoses or ketoses are reducing sugars. They contain free aldehyde or ketone group.
Question 3.
Write two main functions of carbohydrates in plants.
Answer:
Main functions of carbohydrates in plants are :
- Structural material for plant cell walls-For example, the polysaccharides cellulose acts as the chief structural material of the plant cell walls.
- Bio Fuels-Carbohydrates such as glucose, fructose, sugar, starch, glycogen etc. act as biofuels and hence provide energy for the functioning of the living systems.

In the living systems, the polysaccharides such as starch and glycogen are first hydrolysed by enzymes to glucose. This is then transported from one cell to another by cell saps in plants. Oxidation of glucose to CO2 and water by a series of enzyme-catalysed reactions provides energy for the functioning of the cells. - Reserve food materials-Starch is the major food reserve material in plants. It is stored in seeds and acts as the reserve food material for the tiny plant till it is capable of making its own food by photosynthesis.
Question 4.
Classify the following into monosaccharides and disaccharides. Ribose, 2-deoxyribose, maltose, galactose, fructose and lactose.
Answer:
Monosaccharides are ribose, 2-deoxyribose, galactose and fructose. Dissacharides are maltose, lactose.
![]()
Question 5.
What do you understand by the term glycosidic linkage?
Answer:
Disaccharides on hydrolysis with dilute acids or enzymes yield two molecules of either the same or different monosaccharides. The two monosaccharides are joined together by an oxide linkage formed by the loss of water molecule. Such a linkage between two monosaccharide units through oxygen atom is called glycolidic linkage. This glucosidic linkage is also present in polysaccharides like starch and cellulose.
Question 6.
What is glycogen? How is it different from starch?
Answer:
Glycogen-The carbohydrates are stored in animal body as glycogen. It is also known as animal starch because its structure is similar to amylopectin and is rather more highly branched. It is present in liver, muscles and brain. When the body needs glucose, enzymes break the glycogen down to glucose. Glycogen is also found in yeast and fungi. On the other hand, starch is the main storage polysaccharide of plants. It is the most important dietary source for human beings. High context of starch is found in cereals, roots, tubers and some vegetables. It is a polymer of two components-Amylose (15-20%) which is water-soluble and amylopectin (80-85%) which is water-insoluble.
Question 7.
What are the hydrolysis products of (a) sucrose (b) lactose.
Answer:
(a) Sucrose on hydrolysis gives one unit of glucose and one unit of fructose.

(b) Lactose on hydrolysis with dilute acids yields an equimolar mixture of D-glucose and D-galactose.
Question 8.
What is the basic structural difference between starch and cellulose?
Answer:
The basic structural difference between starch and cellulose is of linkage between the glucose units. In starch, there is α-D-glucosidic linkage. Both the components of starch-amylose and amylopectin are polymer of α-D-glucose. On the other hand, cellulose is a linear polymer of β-D-glucose in which C1 of one glucose unit is connected to C4 of the other through β-D-glucosidic linkage.
Question 9.
What happens when D-glucose is treated with the following reagents?
(i) HI,
(ii) Bromine water,
(iii) HNO3.
Answer:
(i) Reaction of D-glucose with HI-
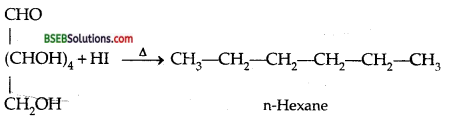
(ii) Reaction of D-glucose with Bromine water-
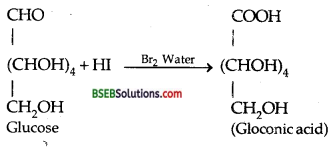
(iii) Reaction of D-glucose with HNO3–
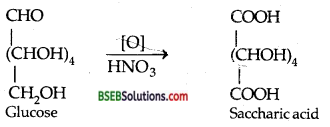
Question 10.
Enumerate the reactions of D-Glucose which cannot be explained by its open-chain structure. ‘
Answer:
The open-chain structure of D-glucose OHC-(CHOH)4– CH2OH fails to explain the following reactions :
(i) Though it contains the aldehyde (- CHO) group, glucose does not give 2, 4-DNT test, Schiff’s test and it does not form the hydrogen sulphite addition product with NaHSO3.
(ii) The pentaacetate of glucose does not react with hydroxylamine (NH2OH) to form the oxime indicating the absence of free – CHO group.
(iii) The formation of two anomeric methyl glucosides by glucose on reaction with CH3OH and dry HCl can be explained in terms of the cyclic structure. Tire equilibrium mixture of α- and β-glucose react separately with methanol in the presence of dry HCl gas to form the corresponding methyl D-glucosides.
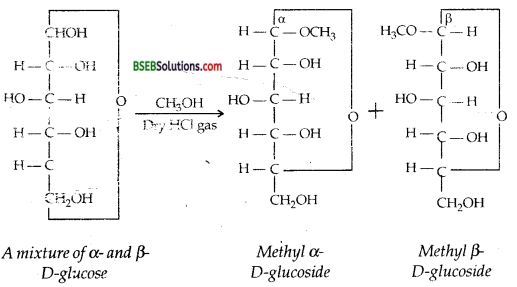
(iv) Like methyl glucosides, glucose pentaacetate also exists in two anomeric forms as explained below:

These pentaacetates do not have a free – OH group at C1 and hence are not hydrolysed in aqueous solution to produce the open-chain aldehydic form and hence do not react with NH2OH to form glucose oxime.
(v) The existence of glucose in two crystalline forms termed as α and β-D-glucose can again be explained on the basis of cyclic structure of glucose and not by its open-chain structure. It was proposed that one of the – OH group may add to – CHO group and form a cyclic hemiacetal structure. It was found that glucose forms a 6-membered ring in which – OH at C – 5 is involved in ring formation. This explains the absence of – CHO group and also existence of glucose in two forms as shown below. These two forms exist in equilibrium with open-chain structure.
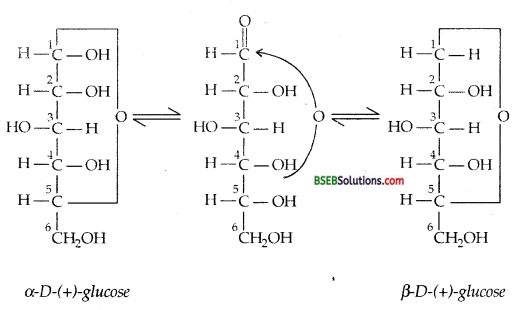
![]()
Question 11.
What are essential and non-essential amino acids? Give two examples of each type?
Answer:
Out of the 20 amino acids required for protein synthesis, human body can synthesize only 10, These 10 amino acids which, the body can synthesize are called non-essential or dispensable amino acids while the remaining ten which the human body cannot synthesize are called essential or indispensable amino acids. These essential amino acids are required for the growth of the body and they must be supplied in the human diet.
Two examples of essential amino acids are valine and phenylalanine. Two examples of non-essential amino acids are glycine and cysteine.
Question 12.
Define the following as related to proteins:
(i) Peptide linkage,
(ii) Primary structure,
(iii) Denaturation.
Answer:
(i) Proteins are the polymers of a-amino acids linked through peptide bond or peptide linkage.
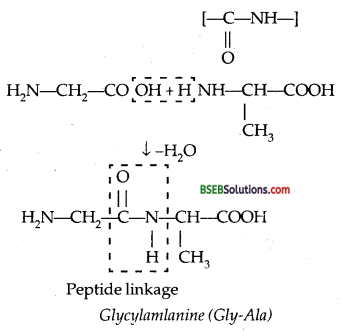
If a third amino acid combines to a dipeptide the product is called a tripeptide. When the number of such amino acids is more than 10, the product is called polypeptide. A polypeptide with more than 100 units of amino acids residues, having molecular mass higher than 10,000 μ is called a protein.
(ii) Primary structure of proteins-Proteins may have one or more polypeptide chains. Each polypeptide in a protein has amino acids linked with each other in a specific sequence and it is this sequence of amino acids that is said to be the primary structure of that protein. Any change in this primary structure, i.e., the sequence of amino acids creates a different protein.
(iii) Denaturation of proteins-When a protein in its native form, is subjected to physical change like change in temperature or chemical change like change in pH, the hydrogen bonds are disturbed. Due to this, globules unfold and helix get uncoiled and protein loses its biological activity. This is called denaturation of proteins. During denaturation 2° and 3° structures are destroyed but 1 ° structure remains intact. The coagulation of egg white on boiling is a common example of denaturation. Another example is curdling of milk which is caused due to the formation of lactic acid by the bacteria present in milk.
Question 13.
What are the common types of secondary structure of proteins?
Answer:
Secondary structure of Proteins-The secondary structure of proteins refers to the shape in which a long polypeptide chain can exist. They are found to exist in two different types of structures viz. α-helix and fl-pleated sheet structure. These structure arise due to the regular folding of tire backbone of the polypeptide drain due to hydrogen bonding between 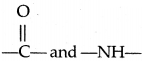 groups of the peptide bond.
groups of the peptide bond.
α-Helix is one of the most common ways in which a polypeptide chain forms all possible hydrogen bonds by twisting into a right-handed screw (helix) with the – NH group of each amino acid residue hydrogen-bonded to the >C = O of an adjacent turn of the helix as shown in Fig. below.
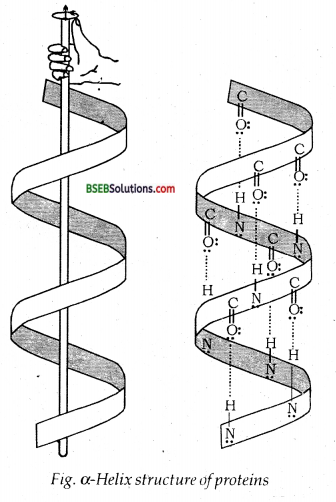
In β-structure, all peptide chains are stretched out to nearly maximum extension and then laid side by side which are held together by intermolecular hydrogen bonds. The structure resembles the pleated folds of drapery and therefore is known as β-pleated sheet.
Question 14.
What type of bonding helps in stabilising the a-helix structure of proteins.
Answer:
α-helix is one of the most common ways in which a polypeptide chain forms all possible hydrogen bonds by twisting into a right-handed screw (helix) with the – NH group of each amino acid residue hydrogen-bonded to the > C = O of an adjacent turn of the helix (as shown in the diagram of answer to Q. 14.13 above).
Question 15.
Differentiate between globular and fibrous proteins.
Answer:
On the basis of molecular structure, proteins have been classified as globular and fibrous proteins. The difference between the two are:
| Globular | Fibrous proteins |
| 1. The polypeptide chain is folded around itself so the entire protein molecule acquires a spherical shape. | 1. These proteins consist of linear thread-like molecules which tend to lie side by side to form fibres. |
| 2. They are water-soluble, as water molecules interact strongly with the polar groups. | 2. Intermolecular forces of attraction are very strong so they are insoluble in water. |
| 3. Globular proteins are very sensitive to small changes in pH and temperature. | 3. Fibrous proteins are stable to moderate changes in temperature and pH. |
Question 16.
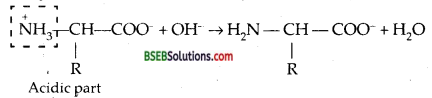
How do you explain the amphoteric behaviour of amino acids?
Answer:
Due to dipolar or Zwitterionic structure, amino acids are amphoteric in nature. The acidic character of the amino acids is due to the N+H3 group while the basic character is due to the COO– group.
![]()
Question 17.
What are enzymes?
Answer:
All biological reactions are catalysed by special catalysts called enzymes. Thus enzymes are biological catalyst. An enzyme facilitates a biochemical reaction by providing alternate lower activation energy pathways thereby increasing the rate of the reaction. Chemically all enzymes are globular proteins. However same enzymes are also associated with same non-protein component called the co-factor for their activity. The enzymes differ from other type of catalyst in being highly specific and selective.
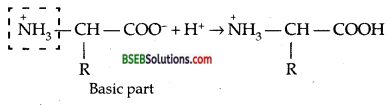

Enzymes, like catalyst are needed only in small quantities.
Question 18.
What is the effect of denaturation on the structure of proteins?
Answer:
Proteins are very sensitive to the action of heat, mineral acids, alkalies etc. On heating or on treatment with mineral acids, soluble forms of proteins such as globular proteins often undergo coagulation Or precipitation to give fibrous proteins which are insoluble in water. This coagulation also results in the loss of the biological activity of the protein. That is why the coagulated proteins so formed are called denatured proteins. Chemically, denaturation does not change the primary structure but brings about changes in the secondary and tertiery structure of proteins.
Question 19.
How are vitamins classified? Name the vitamin responsible for the coagulation of blood.
Answer:
Vitamins are classified into two groups depending upon their solubility in water or fat.
(i) Fat-Soluble Vitamins-Vitamins which are soluble in fat and oils but insoluble in water are kept in this group. These are vitamins A, D, E and K. They are stored in liver and adipose (fat strong tissues).
(ii) Water Soluble Vitamins-B group vitamins and vitamin C are soluble in water so they are grouped together. Water-soluble vitamins must be supplied regularly in diet because they are readily excreted in urine and cannot be stored (except vitamin B12) in our body.
Vitamin K is responsible for the coagulation of blood.
Question 20.
Why are Vitamin A and Vitamin C essential to us? Give their important sources.
Answer:
Deficiency of vitamin A causes Xerophthalmia (hardening of cornea of the eye) and night blindness. So its use is essential to us. It is available in fish liver oil, carrots, butter and milk. Promotes growth and increases resistance to diseases. Vitamin C is very essential to us because its deficiency causes Scurvy (bleeding of gums) and pyorrhea (loosening and bleeding of teeth). Vitamin C increases resistance of the body towards diseases. Maintains healthy skin and helps cuts and abrasions to heat properly. It is soluble in water. It is present in citrus fruits, e.g., oranges, lemons, amla, tomatoes, green vegetables (Cabbage) chillies, sprouted pulses and germinated grains.
Question 21.
What are nucleic acids? Mention their two important functions.
Answer:
Nucleic acids-They constitute an important class of biomolecules which are found in the nuclei of all living cells in the form of nucleoproteins (i.e., proteins containing nucleic acid as the prosthetic group). Nucleic acids are the genetic materials of the cells and are responsible for transmission of hereditary effect from one generation to the other and also carry out the biosynthesis of proteins. Nucleic acids are biopolymers (i.e., polymers present in the living system. The genetic information coded in nucleic acids controls the structure of all proteins including enzymes and thus governs the entire metabolic activity in the living organism.
Two important functions of nucleic acids are :
- Replication-The process by which a single DNA molecule produces two identical copies of itself is called cell division or replication.
- Protein Synthesis-DNA may be regarded as the instrument manual for the synthesis of all proteins present in the cell.
Question 22.
What is the difference between a nucleoside and a nucleotide?
Answer:
Nucleoside-A nucleoside contains only two basic components of nucleic acids, i.e., a pentose sugar and a nitrogenous base. It may be represented as Sugar-base.
Depending upon the type of sugar present, nucleosides are of two types:
- Ribonucleosides and
- Deoxyribonucleosides.
Nucleotides-A nucleotide contains all the three basic components of nucleic acids, i.e., a phosphoric acid group, a pentose sugar and a nitrogenous base. In other words, nucleotides are nucleoside monophosphates.
Depending upon the type of sugar present, nucleotides like nucleosides are of two types:
- Ribonucleotides and
- Deoxynucleotides.
Question 23.
The two strands in DNA are not identical but are complementary. Explain.
Answer:
The two strands in DNA are not identical but are complementary.
We know that in DNA molecule, adenine (A) always pairs with thymine (T) and Cytosine (C) always pairs with guanine (G).
Thus sequence of bases in one strand ATGCTTCA
sequence of bases in the complementary strand TACGAAGT
Thus the sequence of bases in the complementary strand of ATGCTTCA of one strand of DNA molecule is TACGAAGT.
![]()
Question 24.
Write the important structural and functional differences between DNA and RNA.
Answer:
The structural difference between DNA and RNA arenas follows:
| DNA | RNA |
| 1. DNA is a double-stranded helix in which two strands are coiled spirally in opposite directions. | 1. It has a single-stranded structure. |
| 2. The sugar molecule is 2- deoxyribose. | 2. The sugar molecules is ribose. |
| 3. Nitrogenous base uracil is not present. | 3. Nitrogenous base thymine is not present. |
| 4. DNA molecules are very large, their molecules weights may vary from 6 million to 16 million. | 4. RNA molecules are much smaller with molecular weights ranging from 20,000 to 40,000. |
Functional differences between DNA and RNA are as follows :
| DNA | RNA |
| 1. DNA has the unique property of replication. | 1. RNA usually does not replicate. |
| 2. DNA controls the transmission of hereditary effects. | 2. RNA controls the synthesis of proteins. |
Question 25.
What are different types of RNA found in the cell?
Answer:
RNA molecules are of three types and they perform different functions. They are named as messenger RNA (m-RNA) ribosomal RNA (r-RNA), transfer RNA (t-RNA).
Bihar Board Class 12 Chemistry Biomolecules Additional Important Questions and Answers
Very Short Answer Type Questions
Question 1.
Mention two main functions of carbohydrates in plants. (D.S.B. 1997)
Answer:
- Structural material for cell walls and
- bio-fuels.
Question 2.
What is mutarotation? (P.S.B. 1998)
Answer:
Change in specific rotation of an optically active compound in solution with time, to an equilibrium value.
Question 3.
Give one example of denatured protein. (H.S.B. 2001)
Answer:
Cheese.
Question 4.
Sketch the Zwitter ion form of amino acetic acid. A.I.S.B. 2002)
Answer:

Question 5.
What is invert sugar? (H.S.B. 2001; P.S.B 2001)
Answer:
An equimolar mixture of glucose and fructose.
![]()
Question 6.
Name the scientist who was awarded the Nobel prize for ’ the synthesis of Insulin.
Answer:
Frederick Sanger.
Question 7.
What is nucleoside? (P.S.B. 2000)
Answer:
When a nitrogenous base (purine or pyrimidine) is attached to C of sugar (ribose or deoxyribose).
Question 8.
Name the enzyme whose deficiency causes phenyl ketone urea? (H.P.S.B. 2002)
Answer:
Phenylalanine hydroxylase.
Question 9.
Name two different types of RNA molecules found in the cells of organisms? (D.S.B. 2001)
Answer:
mRNA and rRNA.
Question 10.
Why are carbohydrates generally optically active? (A.I.S.B. 1998)
Answer:
Their molecules are chiral because of the presence of many asymmetric carbon atoms.
Question 11.
State the use for the enzyme streptokinase in medicine? (A.I.S.B. 2001)
Answer:
To check heart attacks since it dissolves blood clots formed in coronary artery.
Question 12.
What is colloidon? (H.P.S.B. 2000)
Answer:
A solution of proxy line in ether and alcohol.
![]()
Question 13.
Name the three nucleic acids which are used in protein ” synthesis. (P.S.B. 2000)
Answer:
mRNA, rRNA and tRNA.
Question 14.
What is the efficiency of energy conservation in human body?
Answer:
38%.
Question 15.
If one strand of DNA has the sequence TATCGACEA, write the sequence of bases of mRNA molecule synthesised on it. (D.S.B. 1996)
Answer:
AUAGCUGCU.
Question 16.
Name the central metal atoms present in haemoglobin and Chlorophyll. (H.P.S.B. 2000)
Answer:
Iron (Fe) in haemoglobin and Magnesium (Mg) in chlorophyll.
Question 17.
Name the building blocks of proteins.
Answer:
α-Amino acids.
Question 18.
Name one fibrous and one globular protein. (H.S.B. 2001)
Answer:
Keratin (fibrous); Haemoglobin (globular).
Question 19.
What is a prosthetic group?
Answer:
A prosthetic group is a non-protein portion obtained by hydrolysis of conjugated proteins. Tire main function of the prosthetic group is to control the biological functions of proteins.
Question 20.
Which polysaccharide is stored in the liver of the animals?
Answer:
Glycogen.
Question 21.
Name the vitamin whose deficiency is responsible for the poor coagulation of the blood.
Answer:
Vitamin K.
Question 22.
What does ATP stand for?
Answer:
Adenosine triphosphate.
Question 23.
Name the enzymes present in the saliva of human beings. (P.S.B. 2003)
Answer:
Amylases.
Question 24.
Deficiency of which vitamin causes Scurvy? (P.S.B. 2003)
Answer:
Vitamin C.
Question 25.
Name the vitamins deficiency of which causes (i) rickets, (ii) night blindness. (A.I.S.B. 87, P.S.B. 2003)
Answer:
- Vitamin D,
- Vitamin A.
Question 26.
Deficiency of which vitamin causes beri-beri and pain in joints?
Answer:
Vitamin B1.
Question 27.
Name two important polysaccharides of D-glucose.
Answer:
Starch and Cellulose.
Question 28.
What is a peptide bond? (H.P.S.B. 1997, H.S.B. 2001, P.S.B. 2003)
Answer:
The covalent bond -NH-CO-formed between -NH2 group of one amino acid and -COOH of the other with elimination of a molecule of water is called a peptide bond.
Question 29.
What is hypervitaminoses and avitaminoses? (P.S.B. 2005)
Answer:
Excess intake of vitamins A and D causes hypervitaminoses and multiple deficiencies caused by more than one vitamin is called avitaminosis.
Question 30.
Which is the sugar present in DNA. (A.I.S.B. 2004)
Answer:
Deoxyribose.
![]()
Question 31.
What are oligosaccharides? (H.S.B. 2003)
Answer:
Carbohydrates which on hydrolyses give 2-10 units of monosaccharides are called oligosaccharides. For example sucrose, raffinose, stachyose.
Question 32.
What do you mean by non-essential amino acids? (H.S.B. 2003)
Answer:
Amino acids which the body can synthesise are called non- essential amino acids. For example glycine, alanine and cysteine etc.
Question 33.
What are disaccharides? Give an example. (H.S.B. 2003)
Answer:
Disaccharides on hydrolysis give two units of the same or different monosaccharides. For example sucrose.
Question 34.
How many amino acids occur in almost all proteins. (H.S.B. 2003)
Answer:
20.
Question 35.
What are Polysaccharides? Give examples. (H.S.B. 2003)
Answer:
Polysaccharides are those carbohydrates which give a large number of monosaccharide units on hydrolysis. Starch and cellulose.
Question 36.
Give the structural formula of a nucleotide. (H.P.S.B. 2002)
Answer:
Base-ribose-phosphate or Base-deoxyribose-phosphate.
Question 37.
Why are carbohydrates generally optically active?
Answer:
Because their molecules are chiral due to the presence of many asymmetric carbon atoms.
Question 38.
What are the constituents of starch?
Answer:
Amylose and amylopectin.
Question 39.
What is the secondary structure of proteins?
Answer:
Confirmation which a polypeptide chain assumes as a result of H-bonding.
![]()
Question 40.
Name the nucleic acid which has double helix structure?
Answer:
DNA.
Short Answer Type Questions
Question 1.
How do anomers differ from epimers?
Answer:
Carbohydrates which differ in configuration at the glucosidic; carbon (i.e., C1 in aldoses and C2 in Ketoses) are called anomers. While those which differ in configuration at any carbon atom other than the glucosidic carbon are called epimers. For example α-D-glucose and β-D- glucose are anomers since they differ, in configuration at C1 (glucosidic C) and glucose and mannose are called epimers as they differ in configuration at C2 (other than the glucosidic C atom).
Question 2.
Glucose and Fructose give the same osazone. Explain.
Answer:
During osazone formation, the reaction occurs only at C1 and C2 while the rest of the molecule remains intact. Since glucose and fructose differ from each other only in the arrangement of atoms at C1 and C2, therefore, they give the same osazone.
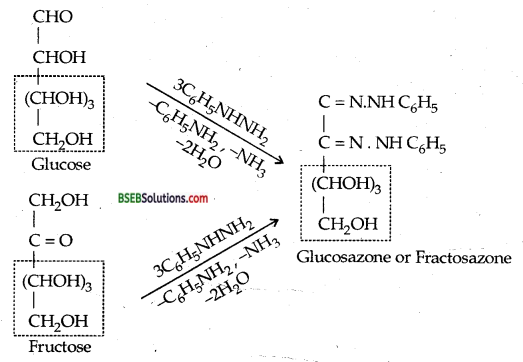
Identical portions are written in boxes.
Question 3.
State differences between primary and secondary structure of proteins. (D.S.B. 2001)
Answer:
The order in which the amino acids are joined in one or more polypeptide chains of a protein is called primary structure. It gives its function and is critical of its biological activity, e.g. – Val – His-Leu- Thr-Pro-Glu-Lys-.
The secondary structure determines the manner in which the protein chain is folded. It arises from planar geometry of the peptide bond and hydrogen bonds between one region of the backbone to another. e.g., α-helix, β-sheets.
Question 4.
What do you mean by nucleoside? Write four functions of RNA. (H.S.B. 2002)
Answer:
A nucleoside contains only two basic components of nucleic acids i.e., a pentose sugar and nitrogenous base.
Functions of RNA:
- RNA controls the synthesis of protein.
- It acts as a catalyst.
- It is the hereditary material in some viruses.
- Sometimes RNA can synthesize RNA and DNA via RNA replication and reverse transcription respectively.
Question 5.
Describe the denaturation of proteins. (A.I.S.B. 2001)
Answer:
(i) Denaturation of Proteins-On heating or on treatment with mineral acids, alkalies etc., soluble forms of proteins such as globular proteins undergo coagulation or precipitation. It results in the loss of the biological activity of the protein, e.g., when milk is heated with lemon juice cheese is formed.
![]()
Question 6.
Draw the Haworth Projection formulae of α-D(+)- Glucopyranose and β-D (+)-Glucopyranose.
Answer:
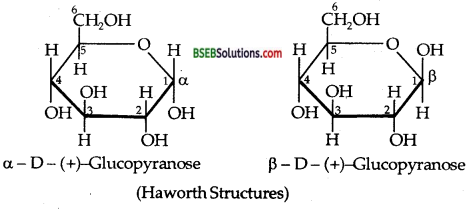
Question 7.
Draw the Haworth projection formulae of α-D(-) fracto faranose and β-D (-) fructofuranose. —
Answer:
The cyclic structures of two anomers of fructose as represented by Haworth structures are given below:

Question 8.
Glycine exists as a Zwitterion but o- and p-aminobenzoic acids do not. Explain.
Answer:
In o- or p-benzoic acids, the lone pair of electrons on the – NH2 group is donated towards the benzene ring. As a result, the acidic character of – COOH group and basic character of – NH2 group decreases. Therefore, the weakly acidic – COOH group cannot transfer to H+ ion to the weakly basic – NH2 group. Thus o- or p- aminobenzoic acids do not exist as Zwitterions.
Question 9.
Define mutation. (P.S.B. 1998, H.P.S.B. 2004, P.S.B. 2005)
Answer:
A mutation may be defined as a chemical change in the sequence of nitrogenous bases along the DNA strands that can lead to the synthesis of proteins with altered amino acid sequences. The changes in DNA molecule can occur spontaneously or may be caused by radiation, chemical changes or viruses.
Question 10.
(i) What is meant by inversion of sugar, (ii) Write down the structure of structure of sugar present in DNA.
Answer:
(i) Sucrose is dextro¬rotatory but on hydrolysis it gives an equimolar mixture of D (+) – glucose and D (-). Fructose which is laevorotatory. This change in specific rotation from dextrorotation to laevorotation is called inversion of sugar.
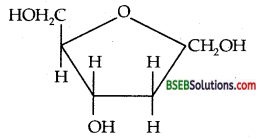
(ii) The sugar present in DNA is deoxyribose whose structure is given along with.
![]()
Long Answer Type Questions
Question 1.
(a) What are enzymes? How do they act?
(b) What are Vitamins. How are they classified.
Answer:
(a) Enzymes are biological catalysts. Almost all enzymes are globular proteins. Enzymes are very specific for a particular reaction and for a particular substrate. They are generally named after the compound or class of compounds upon which they work. For example, the enzyme that catalyses hydrolysis of maltose into glucose is named as maltose.

Sometimes enzymes are also named after the reaction, where they are used. For example, the enzymes which catalyse the oxidation of one substrate with simultaneous reduction of another substrate are named as oxidoreductase enzymes. The ending of the name of an enzyme is -ase.
Enzymes are needed only in small quantities for the progress of a reaction. Similar to the action of chemical catalysts, enzymes are said to reduce the magnitude of activation energy. For example, activation energy for acid hydrolysis of sucrose is 6.22 kj mol-1, while the activation energy is only 2.15 kj mol-1 when hydrolysed by the enzyme, sucrase.
(b) Vitamins: Vitamins are the organic compounds required in diet in small amounts to perforin specific biological functions for normal maintenance of optimum growth and health of the organism. Vitamins are designated by alphabets A, B, C, D, etc. Some of them are further named as sub-groups e.g. B1, B2, B6, B12, etc. Excess of vitamins is also harmful and vitamin pills should not be taken without the advice of doctor.
Vitamins are classified into two groups depending upon their solubility in water or fat
(i) Fat-soluble vitamins: Vitamins which are soluble in fat and oils but insoluble in water are kept in this group. These are vitamins A, D, E and K. They are stored in liver and adipose (fat-storing) tissues.
(ii) Water-soluble vitamins: B group vitamins and vitamin C are soluble in water so they are grouped together. Water-soluble vitamins must be supplied regularly in diet because they are readily excreted in urine and cannot be stored (except vitamin B12) in our body.
![]()
Question 2.
What are nucleic acids? What are their chemical composition? What are the primary and secondary structures of nucleic acids.
Answer:
Nucleic acids-The particles in the nucleus of cell, responsible for heredity, are called chromosomes which are made up of proteins and other type of biomolecules called nucleic acids. They are mainly of two types:
- Deoxyribonucleic acid (DNA)
- Ribonucleic acid (RNA)
Since nucleic acids are long-chain polymers of nucleotides, so they are also called polynucleotides.
Chemical composition of Nucleic acids—Complete hydrolysis of DNA (or RNA) yields a pentose sugar, phosphoric acid and nitrogen-containing heterocyclic compounds called bases. In DNA molecules the sugar part is β-D-2-deoxy ribose whereas RNA molecule, it is β-D-ribose.
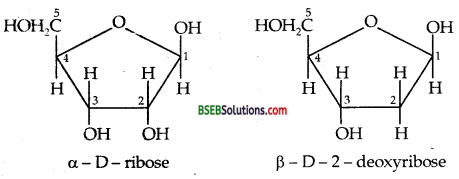
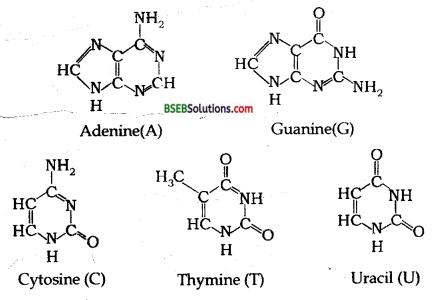
DNA contains four bases viz. adenine (A), guanine (G), cytosine (C) and thymine (T). RNA also contains four bases, the first three bases are same as in DNA but the fourth one is uracil (U).
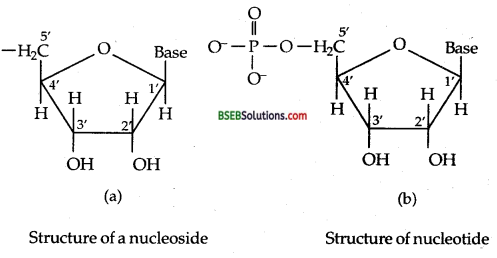
Structure of Nucleic acids-A unit formed by the attachment of a base to V position of sugar is known as nucleoside. In nucleoside, the sugar carbons are numbered as 1′, 2′ 3′ etc. in order to distinguish these from the bases [Fig. (a) below]. When nucleoside is linked to phosphoric acid at 5-position of sugar moiety, we get a nucleotide [Fig. (b) below].
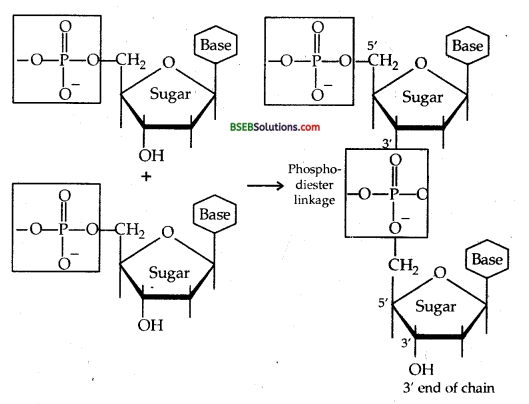
Nucleotides are joined together by phosphodiester linkage between 5′ and 3′ carbon atoms of the pentose sugar. The formation of a typical dinucleotide is given along with.
A simplified version of nucleic acid chain is as shown below. Base Base Base
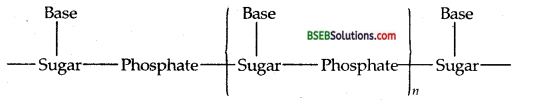
Information regarding the sequence of nucleotides in the chain of a nucleic acid is called its primary structure. Nucleic acids have a secondary structure also. James Watson and Francis Crick gave a double-strand helix structure for DNA (Fig.). Two nucleic acid chains are wound about each other and held together by hydrogen bonds between pairs of bases. The two strands are complementary to each other because the hydrogen bonds are formed between specific pairs of bases. Adenine forms hydrogen bonds with thymine whereas cytosine forms hydrogen bonds with guanine.
In secondary structure of RNA, helices are present which are only single-stranded. Sometimes they fold back on themselves to form a double helix structure. RNA molecules are of three types and they perform different functions. They are named as messenger RNA (m-RNA), ribosomal RNA (r-RNA) transfer RNA (t-RNA).
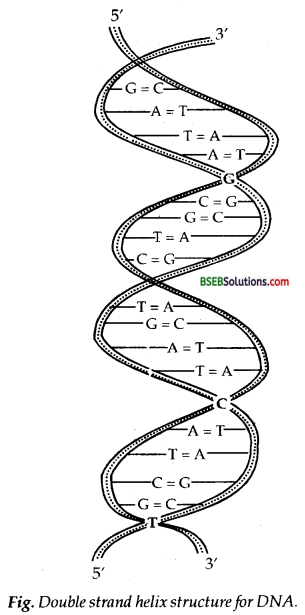
Fig. Double strand helix structure for DNA.