BSEB Bihar Board Class 9 Science Solutions Chapter 1 Matter in Our Surroundings
Bihar Board Class 9 Science Solutions Chapter 1 Matter in Our Surroundings Textbook Questions and Answers, Additional Important Questions, Notes.
Bihar Board Class 9 Science Chapter 1 Matter in Our Surroundings InText Questions and Answers
Intext Questions (Page 3)
Question 1.
Which of the following are matter :
Chair, air, love, smell, hate, almonds, thought, cold, cold drink, smell of perfume ?
Answer:
Matter :
Chair, air, almonds, cold drink.
Not Matter :
Love, smell, hate, thought, cold, smell of perfume.
Question 2.
The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Answer:
The rate of diffusion is very high in case of gases. The particles of hot sizzling food mixes with the particles of air and reaches us several metres away. In case of cold food the food particles do not mix with the particles of air easily. The temperature increase the rate of diffusion. Hence we have to go close to it to get the smell.
![]()
Question 3.
A diver is able to cut through water in a swimming pool. Which property of matter does this observation show ?
Answer:
This shows that liquids are compressible. The intermolecular space between the particles of liquids is greater as compared tothe solids. Hence when the diver dives into water the particles of water get compressed.
Question 4,
What are the characteristics of the particles of matter ?
Answer:
(i) Particles of matter are continuously moving, that is they possess kinetic energy.
(ii) Particles of matter intermix on their own with each other. They do so by getting into the spaces-between the particles.
page 6
Question 1.
The mass per unit volume of a substance is called density. (density = mass/volume). Arrange the following in order of increasing density air,exhaust from chimneys, honey, water, chalk, cotton and iron.
Answer:
Air, exhaust from chimnies, water, honey, cotton, chalk, iron.
Question 2.
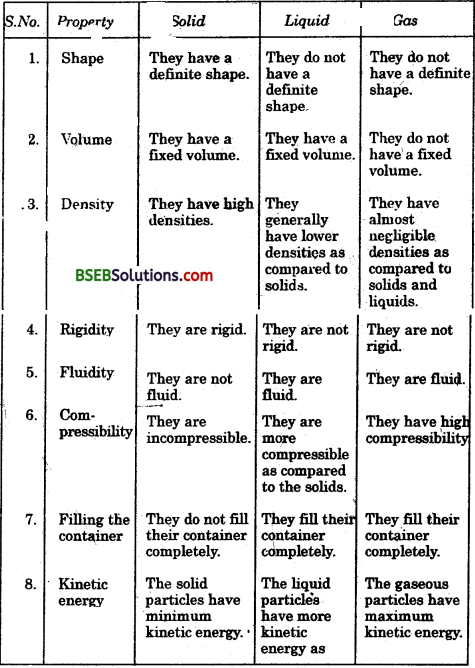
(a) Tabulate the differences in the characteristics of states of matter. 3
(b) Comment upon the following : rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy and density.
Answer:
Question 3.
Give reasons
(a) A gas fills completely the vessel in which it is kept.
(b) A gas exerts pressure on the walls of the container.
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood we need a karate expert.
Answer:
(a) A gas ifils completely the vessel in.which it is kept due to large intermolecular space between the gaseous particles and high kinetic energy of the particles. Moreover the gas is fluid.
(b) The kinetic energy of the particles in gaseous state is the maximum. Particles move about randomly at high speed. Due to random movement the particles hit each other and also the, walls of the container. The pressure exerted by the gajs is because of this force exerted by gas particles per unit area of the container.
(c) A wooden table should be called a solid because it ^ has a definite shape, distinct boundary and fixed volume.
(d) The particles of air have weak intermolecular force of attraction. They also have a large intermolecular space between them. As a result we can easily move our hand in air. In case of a solid block of wood or brick the particles are closely packed having a strong – intermolecular force of attraction and very less intermolecular space. Hence, it is difficult to move our ” hand through it.
![]()
Question 4.
Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why. Answer:
Ice has greater volume. Hence, it has less
density ![]() Density as compared to water thus it floats on water
Density as compared to water thus it floats on water
PAGE 6
Question 1.
Cohvert the following temperature to Celsius scal.
(a) 300 K
(b) 573 K.
Answer:
(a) 300 K
= (300 – 273)°C
= 27°C
(b) 573 K
= (573 – 273)°C
= 30Q°C ,
Question 2.
What is the physical state of water at:
(a) 250°C
(b) 100°C ?
Answer:
(a) At 250°C the physical state of water is gas.
(b) At 100°C the physical state of water changes from liquid to vapour. –
Question 3.
For any substance, why.does the temperature remain constant during the change of state ?
Answer:
The temperature remains constant during the change of state because the head gets used up in changing the state by overcoming the forces of attraction between . the particles. The excess heat is absorbed by the particles in the form of latent heat.
Question 4.
Suggest a method to liquefy atmospheric gases.
Answer:
The air is compressed by increasing the pressure and is then cooled by decreasing the temperature to get liquid air.
Page 10
Question 1.
Why does a desert cooler cool better on a hot dry day ?
Answer:
A cooler cools better on a hot dry day because the rate of evaporation is directly proportion he temperature and inversely proportional to humidny. Air around us cannot hold more than a definite amount of water vapour at a given temperature. On a hot dry day the rate of evaporation increases (air can hold more water vapour) thus causing a cooling effect.
Question 2.
How does the water kept in an earthen pot (matka) become cool during summer ?
Answer:
An earthen pot has a large number of extremely small pores. Water seeps out through them and evaporates from the surface of the pot, thereby cooling the whole system. The latent heat required for evaporation is taken from the water in the pot. As a result water becomes cool.
![]()
Question 3.
Why does our palm feel cold when we put some acetone or petrol or perfume on it ?
Answer:
(i) When we put acetone on our palm the particles gain energy from the palm or surroundings and leave Ans. The water vapour present in the air on coming in contact with the cold beaker of ice loses energy and , gets the surface of the palm giving a cool feeling.
(ii) When we put acetone on our palm the particles gain energy from the palm or surroundings and leave the surface of the palm giving a cool feeling.
Question 4.
Why are we able to sip hot tea or milk -foster from a saucer rather than a cup ?
Answer:
We are able to sip hot tea faster from a saucer rather than a cup because a saucer has a greater surface area. As a result rate of evaporation increases and cools the tea.
Question 5.
What type of clothes should we wear in summer ?
Answer:
We should wear cotton clothes during summer season. I)uring summer we perspire more because of the mechanism of our body to keep us cool. During evaporation the particles at the surface of the liquid gain energy from the surroundings and change into vapour. The heat energy equal to the latent heat of vaporization is absorbed from the body leaving the body cool. Cotton being a food absorber of water helps in absorbing the sweat; and exposing it to |he atmosphere for easy
Bihar Board Class 10 Science Chapter 1 Matter in Our Surroundings Textbook Questions and Answers
Question 1.
Convert the following temperatures to the Celsius scale.
(a) 300 K
(b) 573 K.
Answer:
(a) 300 K
= (300 – 273)°C
= 27°C
(b) 573 K
= (573 – 273)°C
= 300°C
Question 2.
Convert the following temperatures to Kelvin scale.
(a) 25°C
(b) 373°C.
Answer:
(a) 25°C
= (25 + 273) K
= 298 K
(b) 373°C
= (373 +273) K
= 646K
Question 3.
Give reason for the following observations.
(a) Naphthalene balls disappear with time without leaving any solid.
(b) We can get the smell of perfume sitting severaj metres away.
Answer:
(a) Naphthalene baflls disappear with time without
they directly change into vapour without passing through the liquid state.
(b) The particles of perfume mix on their own with the particles of air around us and spread out. Due to this spreading of the particles we can get the smell of peirfume sitting at a distance.
![]()
Question 4.
Arrange the following substances in increasing order of forces of attraction between the particles – water, sugar, oxygen.
Answer:
The intermolecular force of attraction is least in gas, followed by liquid and maximum in gas. Therefore, it is least in oxygen followed by water and maximum in sugar.
Question 5.
What is the physical state of water at
(a) 25°G
(b) 0°C
(c) 100°C ?
Answer:
(a) 25°C – Liquid
(b) 0°0 – Solid
(c) 100°C – liquid to vapour.
Question 6.
Give two reasons to justify –
(a) water at room temperature is a liquid.
(b) an iron almirah is a solid at room , temperature.
Answer:
(a) Water at room temperature is a liquid is because it does not have a fixed shape. It takes the shape of the container where it is poured. Secondly it can flow easily, hence it is not rigid but a fluid.
(b) An iron almirah is a solid because it has a definite $hape and a fixed volume. It does riot flow, hence it is rigid.
Question 7.
Why is ice at 273 K more effective in cooling than water at the same temperature ?
Answer:
The particles in ice at 273 K have less energy as rcotnpared to the particles of water at 273 K. Hence ice trikes padre heat energy from the surroundings and thus causes anore cooling than water at the same temperature.
Question 8.
What produces more severe burns, boiling \ water pr steam ?
Answer:
The particles in steam at 373 K have more energy than toater at the same temperature. This is , because particles in steam have absorbed jextra enrirgy in the form of latent heat of vaporisation. So when steam comes in contact with skin, it releases more heat than the boiling water would do at the time temperature. As/ a result steam at 373 K causes more severe burns than! boiling water at the same temperature.
Question 9.
Name A, B, C, D, E hnd F in the following diagram showing change in its state.

Answer:
A – Fusion
B – Vaporisation
C – Condensation
D – Solidification
E – Sublimation
F – Sublimation.
Bihar Board Class 10 Science Chapter 1 Matter in Our Surroundings Textbook Activities
Activity 1.1
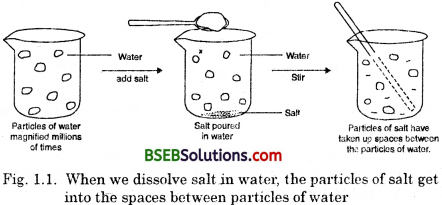
- Take a 100 mL beaker.
- Fill half the beaker with water and mark the level of water.
- Dissolve some salt/sugar with the help of glass rod.
- Observe any change in the water level.
(i) What do you think has happened to the salt ?
(ii) Where does it disappear ?
(iii) Does the level of water change ?
Answer:
(i) The salt dissolves in water (i.e., it spreads out throughout water).
(ii) When we dissolve salt and water the particles of salt goes into the intermolecular space of water (i.e., the space between the particles of matter).
(iii) No, the level of water will not change.
![]()
Activity 1.2
- Take 2-3 crystals of potassium permanganate and dissolve them in 100 mL of water.
- Take out approximately 10 mL of this solution and put it into 90 mL of clear water.
- Take out 10 mL of this solution and put it into another 90 mL of clear water.
- Keep diluting the solution like this 5 to 8 times.
(i) Is the water still coloured ?
(ii) What can you conclude from the above experiment ?

Answer:
(i) Yes, the water is still coloured. A few crystals of potassium permanganate can colour a large volume of water (about 1000 1). (ii) We conclude that there must be millions of tiny particles in just one crystal of potassium permanganate.
Activity 1.3
- Put an unlit incense stick in a corner of your class.
- Now light the incense stick.
(i) How close do you have to go near it so as to get its smell ?
(ii) What happens ? Do you get the smell sitting at a distance ?
(iii) Name the phenomena. When does it increase ?
(iv) Record your observations.
Answer:
(i) When the incense stick is not lighted, we have to go very close to it to get the smell.
(ii) When the incense stick is lighted the particles of perfume in the incense stick mix on their own with the particles of air around us and spread out. Due to this spreading of the particles we can get the smell sitting at a distance.
(iii) This phenomena is called diffusion. (The intermixing of particles of two substances on their own is called diffusion). The diffusion increases on heating.
(iv) Student do it yourself.
Activity 1.4
- Take two glasses/beakers filled with water.
- Put a drop of blue or red ink slowly and carefully by the sides of the first beaker and honey in the same way in the second beaker.
- Leave them undisturbed in your home or in a corner in the class.
- Record your observations.
(i) What do you observe immediately after adding the ink drop ?
(ii) What do you observe immediately after adding a drop of honey ?
(iii) How many hours or days does it take for colour of ink to spread evenly throughout the water spread evenly ?
(iv) What does this suggest about the particles ?
(v) How does diffusion vary with density of liquid ?
Answer:
(i) The particles of ink intermixes with the particles of water.
(ii) The particles of honey settles down at the bottom of the glass and slowly intermixes with the particles of water.
(iii) It takes longer time for honey as compared to ink to spread evenly. (When they are stirred.)
(iv) This suggests that honey being more viscous takes longer time to diffuse, i.e., the particles of honey are close to each other.
(v) Diffusion is inversely proportional to the density of the liquid.
![]()
Activity 1.5
- Drop a crystal of copper sulphate or potassium permanganate into a glass of hot water and another containing cold water. Do not stir the solution. Allow, the crystals to settle at the bottom.
- Observe and report.
(i) What do you observe just above the solid crystal in the glass ?
(ii) What happens as the time passes ?
(iii) What does this suggest about the particles of solid and liquid ?
(iv) Does the rate of mixing change with temperature ? Why and How ?
Answer:
(i) The solid crystal starts spreading out colour in the glass. (Pink in case of potassium permanganate and blue in case of copper sulphate.)
(ii)As time passes more and more colour is given out and the water becomes coloured.
(iii) This suggests that the particles of solid goes into the intermolecular space of the liquid i.e., diffusion takes place of solid into liquid.
(iv) Yes, the rate of diffusion changes wi;h temperature. It is directly proportional to the temperature. As the temperature increases the intermolecular force of attraction decreases, the solid particles gain more kinetic energy and easily diffuse into the liquid. ”
Activity 1.6
- Play this game in the field-make four groups and form human chains as suggested.
- The first group should hold each other from the back and lock arms like Bihu dancers (Fig. 1.3).

- The second group should hold hands to form a human chain.
- The third group should form a chain by touching each other with only their finger tips.
- Now the fourth group of students should run v around and try to break the three human chains one by one into as many small groups as possible.
(i) Which grpup was the easiest to break ? Why ?
(ii) If we consider each student as a particle of matter, then in which group the particles held each other with the maximum force ?
Answer:
(i) The third group was the easiest to break. This is because they are applying the least force to hold each other.
(ii) In the first group the particles are held each other with the maximum force.
Activity 1.7
- Take an iron nail, a chalk piece and a rubber band.
- Try breaking them by hammering, cutting or stretching.
In-which of the above three substances do you think the particles are held together with greater force ?
Answer:
In an iron nail the particles are held together with greater force.
![]()
Activity 1.8
- Give reasons for the following observation :
- Open a water tap, try breaking the stream of water with your fingers.
(i) Were you able to cut the stream of water ?
(ii) What could be the reason behind the stream of water remaining together ?
Answer:
(i) No, we were not able to break the stream of water.
(ii) The stream of water remained together due to a force which kept the water particles together.
Activity 1.9
- Collect the following articles-a pen, a book, a . needle and a piece of thread.
- Sketch the shape of the above articles in your notebook by moving a pencil around them.
(i) Do all these have a definite shape, distinct boundaries and a fixed volume ?
(ii) What happens if they are hammered, pulled or dropped?
(iii) Are these capable of diffusing into each other ?
(iv) Try compressing them by applying force. Are you able to compress them ?
Answer:
(i) Yes, all these have a definite shape, distinct boundaries and a fixed volume.
(ii) If they are hammered, pulled or dropped with a heavy force they may break but it is difficult to change their shape, so they are rigid.
(iii) No, they are not capable of diffusing into each other.
(iv) They have negligible compressi-bility.
Activity
- Write something on one corner of the black board in your class.
- Leave it uncleaned for ten to fifteen days or do not clean the board before going for your autumn break.
(i) Does it become difficult to clean the board afterwards ?
(ii) Think of a reason as to why this happens.
Answer:
(i) Yes, it becomes difficult to clean the blackboard. .
(ii) This happens because the particles of chalk diffuse into the particles of the blackboard.
![]()
Activity 1.10
- Collect the following
(а) water, cooking oil, milk Juice, a cold drink.
(b) containers of different shapes. Put a 50 mL” mark on these containers using a measuring cylinder from the laboratory.
(i) What will happen if these liquids are spilt on the floor ?
(ii) Measure 50 mL of any one liquid and tran sfer it into different containers one by one. Does the volume remain the same ?
(iii) Does the shape of the liquid remain the same ?
(iv) When you pour the liquid from one container into another, does it flow easily ?
Answer:
(i) If,these liquids are spilt on the floor they will flow and change shape, so they are not rigid but can be called fluid.
(ii) Yes, the volume will remain the same.
(iii) No, the shape of the liquid does not remain the same.
(iv)Yes, the liquid flows easily.
Activity 1.11
- Take three 100 mL syringes and close their nozzle by rubber corks as shown in Fig. 1.4.
- Remove the pistons from all the syringes.
- Leaving one syringe untouched, fill water in the second and pieces of chalk in the third.
- Insert the pistons back into the syringes. You may apply some vaseline on the pistons before inserting them into the syringes Fig. 1.4. Demonstrating compressi- for their smooth Mity of a gas movement.

- Now try to compress the content by pushing the piston in each syringe.
(i) What do you observe ? In which case the piston was easily pushed in ?
(ii) What do you infer from your observation ?
Answer:
- The piston was easily pushed in the syringe which was filled with air.
- We infer that gases are highly compressible as compared to liquids.
Activity 1.12
- Take about 150 g of ice in a beaker and I suspend a laboratory thermometer so that its bulb is in contact with the ice, as in Fig. 1.6.
- Start heating the beaker on a low flame.
- Note the temperature when the ice starts melting.
- Recod your observations for this conversion of solid to liquid state.
- Now.put a glass rod in the beaker and heat while stirring till the water starts boiling.
- Keep a careful eye on the thermometer reading till most of the water has vaporised.
- Record your observations for the conversion of water in the liquid state to the gaseous state.

(i) What conclusion can you draw ?
Answer:
(i) Ice will start melting at 0°C (273.16 K).
(ii) When all the ice converts into water the temperature still remains 0°C (273.16 K).
(iii) When liquid water is heated, its temperature gradually increases until it starts boiling. Water boils at 100°C under normal pressure.
(iv) After the liquid water has started boiling (at 100°C), its temperature becomes constant at 100°C even when heating is continued. The temperature remains constant until the whole of water gets vapourised.
(v) We can conclude that the temperature remains constant during change of state.
![]()
Activity 1.13
- Take some camphor or ammonium chloride. Crush it and put in a china dish.
- Put an inverted funnel over the china dish.
- Put a cotton plug on the stem of the funnel, as shown in Fig. 1.7
- Now, heat slowly and observe.
img
(i) What do you infer from the above activity ?
Answer:
(i) We will observe that camphor or ammonium chloride will directly change into vapour.
(ii) We infer that there are certain solids which
directly change into gaseous state without changing into the liquid state.
Activity 1.14
- Take 5 mL of water in a test tube and keep it near a window or under a fan.
- Take 5 mL of water in an open china dish and keep it near a window or under a fan.
- Take 5 mL of water in an open china dish and keep it inside a cupboard or on a shelf in your class.
(i) Record the room temperature.
(ii) Record the time or days taken for the evaporation process in the above cases.
(iii) Repeat the above three steps of activity on a rainy day and record your observations.
(iv) What do you infer about the effect of temperature, surface area and wind velocity (speed) on evaporation?
Answer:
(i) The room temperature will vary from season to season.
(ii) TheAnne for evaporation will depend upon the season.
(iii) The rate of evaporation will be slow during rainy season as compared to the summer.
(iv) The rate of evaporation is directly proportional to ihe teniporature, surface area and wind velocity.
it is inversely proportional to the humidity.
![]()
Bihar Board Class 9 Science Chapter 1 Matter in Our Surroundings Additional Important Questions and Answers
Very Short Answer Type Questions
Question 1.
What is matter ?
Answer:
Anything that occupies space and has mass / is called matter.
Question 2.
Give two examples of master ?
Answer:
Book, table.
Question 3.
Give two examples Which are not matter.
Answer:
Fear, love.
Question 4.
What name was given to the five basic, elements of matter classified by the early Indian philosophers ?
Answer:
The Panch Tatva.
Question 5.
Name the “Panch Tatva.”
Answer:
Air, Earth, Fire, SkyandWater.
Short Answer Type Questions
Question 1.
Write the characteristics of solids.
Answer:
- Solids have a definite shape and a fixed volume.
- Solids have distinct boundaries. They do not fill their container completely.
- They are rigid.
- They are incompressible.
- They have high densities.
- They do not flow.
Question 2.
Define volume. Write its common unit and SI unit.
Answer:
- The space occupied by a substance is called the volume
- The common unit of volume is litre.
- The SI unit of volume is cubic metre (m3).
Question 3.
Define density. Write its formula and unit.
Answer:
The mass per unit volume of a substance is chlled density.
![]()
The unit of density is kg/m3.
Question 4.
(i) What happens when the intermolecular force of attraction is very strong ?
(ii) What happens when the intermolecular space is very less ?
(iii) Although sponge is a solid, it can be compressed. Why ?
Answer:
(i) When the intermolecular force of attraction is very strong the particles of matter are closely packed ( and it takes a solid form. These forces of attraction keep the particles of a solid tightly bound in place. Thus,’ solids have a definite shape
(ii) When the intermolecular space is very less as in ‘ solids the particles cannot be compressed.
(iii) A sponge can be compressed because it has minute holes in which air is trapped. When we press it, the air is expelled out.
Long Answer Type Questions
Question 1.
What type of clothes should we wear during summer season ? Why ?
Answer:
(i) We should wear cotton clothes during summer season.
(ii) During summer we perspire more because of the mechanism of our body to keep us cool. During evaporation the particles at the surface of the liquid gain energy from the surroundings and change into vapour. The heat energy equal to the latent heat of vaporization is absorbed from the body leaving the body cool. Cotton being a good absorber of water helps in absorbing the sweat and exposing it to the atmosphere for easy evaporation.