BSEB Bihar Board Class 9 Science Solutions Chapter 2 Is Matter Around Us Pure
Bihar Board Class 9 Science Solutions Chapter 2 Is Matter Around Us Pure Textbook Questions and Answers, Additional Important Questions, Notes.
Bihar Board Class 9 Science Chapter 2 Is Matter Around Us Pure InText Questions and Answers
Intext Questions
Page 15
Question 1.
What is meant by a pure substance ?
Answer:
The substances which always have the same colour, taste or texture at a given temperature and pressure are pure substances.
Question 2.
List the points of differences between homogeneous and hetero-geneous mixtures.
Answer:
Homogeneous mixture :
- A homogeneous mixture has a uniform composi-tion throughout its mass.
- It has no visible boundaries of separation between the various consti-tuents.
Heterogeneous mixture :
- A heterogeneous mixture does not have a uniform composition throughout its mass.
- It has visible boundaries of separation between the various consti-tuents.
Page 18
Question 1.
Differentiate between homogeneous and heterogeneous mixtures with examples.
Answer:
Homogeneous mixture :
(i) A mixture of salt in water (called salt solution) is a homogeneous mixture because all the parts of salt solution have the same salt-water composition and appear to be equally salty.
(ii) There is no visible boundary of separation between salt and water particles in a salt solution.
![]()
Heterogeneous mixture :
(i) A mixture of sand and iron filings is a heterogeneous mixture because different parts of this mixture will have different sand- iron filings composition.
(ii) There is a visible boundary of separation between iron filings and sand particles.
Question 2.
How are sol, solution and suspension different from each other ?
Answer:
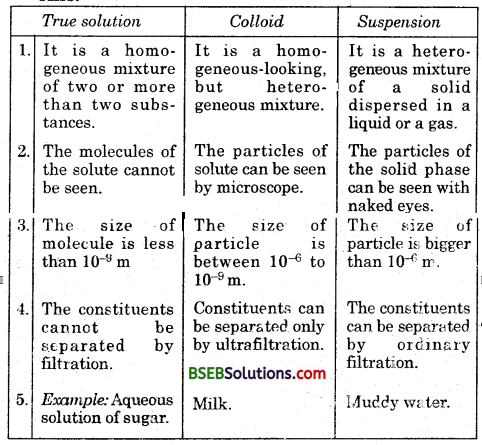
Question 3.
To make a saturated solution, 36 g of sodium chloride is dissolved in 100 g of water at 213 K. Find its concentration at this temperature.
Answer:
Mass of solute (salt) = 36g
Mass of solvent (water) = 100 g
Mass of solution = Mass of solute + Mass of solvent
= 36 g + 100 g = 136 g
Mass percentage of solution
![]()
\(\frac {36}{136}\) x 100 = 26.4
![]()
Page 24
Question 1.
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than 25°G),which are miscible with each oilier.
Answer:
A mixture of kerosene and petrol can be separated by fractional distillation.
Question 2.
Name the technique to separate
- butter from curd.
- salt from sea-water.
- camphor from salt.
Answer:
- Butter from curd can be separated by the process of centrifugation.
- Salt is separated from sea-water by the process of evaporation and crystallization.
- Camphor from salt can be separated by the process of sublimation.
Question 3.
What type of mixtures are separated by the >. technique of crystallisation ?
Answer:
Crystallisation is used to separate a mixture of solid containing impurities and a liquid. Crystallisation separates a pure solid in the form of its crystals from a solution.
Page 24
Question 1.
Classify the following as chemical or physical change :
- Cutting of trees.
- Melting of butter in a pan.
- Rusting of almirah.
- Boiling of water to form steam.
- Passing of electric current through water and the water breaking down into hydrogen and oxygen gases.
- Dissolving common salt in water.
- Making a fruit salad with raw fruits.
- Burning of paper and wood.
Answer:
- Physical change.
- Physical change.
- Chemical change.
- Physical-change.
- Chemical change.
- Physical change.
- Physical change, but if salt is added to the fruit salad then it will be a chemical change.
- Chemical change.
![]()
Question 2.
Try segregating the things around you as pure substances or mixtures.
Answer:
Mixtures – Soil, air, milk, stainless steel utensils, etc.
Pure substance – Distilled water, glucose, iron rod, mercury inside a thermometer, etc.
Bihar Board Class 9 Science Chapter 2 Is Matter Around Us Pure Textbook Questions and Answers
Question 1.
Which separation techniques will you apply for the separation of the following :
(a) Sodium chloride from its solution in water.
(b) Ammonitim chloride from a mixture containing sodium chloride and ammonium chloride.
(c) Small pieces of metal in the engine oil of a car.
(d) Different pigments from an extract of flowrer petals.
(e) Butter from curd.
(f) Oil from water.
(g) Tea leaves from tea.
(h) Iron pins from sand.
(i) Wheat grains from husk.
(j) Fine mud particles suspended in water.
Answer:
(a) Evaporation.
(b) Sublimation.
(c) Filtration.
(d) Fractional distillation.
(e) Chromatography.
(f) Centrifugation.
(g) Separating funnel.
(h) Filtration.
(i) Magnetic separation.
(j) Winnowing.
![]()
Question 2.
Write the steps you would use for making tea. Use the words—solution, solvent, solute, dissolve, soluble, insoluble, filtrate and residue.
Answer:
Step 1 : Boil some water (solvent) in a pan.
Step 2 : Put some tea leaves (solute) in a tea pot.
Step 3 : Pour the boiling water into the pot and let it soak for a few minutes. It will form a solution.
Step 4 : Put sugar (solute) into a cup.
Step 5 : Stir the solution in the tea pot.
Step 6 : Use a strainer and pour the solution into the cup.
Put two teaspoons of milk. Remove the strainer and stir with a spoon. The tea is ready. The tea leaves (residue) will be left on the strainer while tea (filtrate) will pass thro’ugh the solution. Sugar and milk are soluble solutes while tea leaves are insoluble solute.
Question 3.
Pragya tested the solubility of three different substances at different temperatures and collected the data as given below (results are given in the following table, as grams of substance dissolved in 100 grams of water to form a saturated solution.)

(a) What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313 K ?
(b) Prag.ya makes a saturated solution of potassium chloride in water at 353 K and leaves the solution to cool at room temperature. What would she observe as the solution cools ? Explain.
(c) Find the solubility of each salt at 293 K. Which salt has the highest solubility at this temperature ?
(d) What is the effect of change of temperature on the solubility of a salt ?
Answer:
The maximum amount of a solute which can be dissolved in 100 grams of a solvent is known as the solubility of that solute in that solvent (at a specified temperature and pressure).
(a) At 313 K, 100 g of water can dissolve a maximum of 62 g of potassium nitrate.
At 313 K, 50 g of water will be able to dissolve a maximum of 31 g of potassium nitrate.
(b) Room temperature is taken as 20°G (approximately).
30°C – (20 + 273) K = 293 K
![]()
At 353 K, 100 g of water can dissolve a maximum of 54 g of potassium chloride whereas at 293 K, 100 g of water can dissolve a maximum of 35 g of potassium chloride. The solubility of solids in liquids decreases with the decrease in f temperature. Hence, powders of potassium chloride will start settling down.
(c) At 293 K,
Solubility of potassium nitrate = 32g
Solubility of sodium chloride = 36 g
Solubility of potassium cliloride = 35 g
Solubility of ammonium chloride =24g.
At 293 K, sodium chloride has the maximum solubility.
(d) (i) The solubility of solids il|..liquids increases on. increasing the temperature and decreases on decreasing the temperature.
(ii) The solubility of gases in liquids decreases ori increasing the temperature and increases on decreasing the temperature. _
Question 4.
Explain the following giving examples :
(a) Saturated solution
(b) Pure substance
(c) Colloid
(d) Suspension.
Answer:
(a) Saturated .solution – At atty particular temperature, a solution that has dissolved as much solute as it is capable of dissoving it, is said to be saturated solution. For example, heat about 100 ml of water in a beaker to about 25°C and dissolve sodium chloride in this water gradually, until no more sodium chloride goes into the v solution. Maintain the solution at 25°C in a water bath. The supernatent solution is the saturated solution of sodium chloride in water at 25°C.
(b) The substances which always have the same colour, taste or texture at a given temperature and pressure are pure substances. For example, distilled water.
![]()
(c) A heterogeneous mixture in which the size of the solute particles is between lnm (10-9m) and 100 nm (107m) is known as colloidal solution. For example; Milk, Gum, Jelly.
(d) A suspension is a heterogeneous mixture in which the solute particles do not dissolve but remain suspended throughout the bulk of the medium. For example, muddy wtffer, saw dust – water mixture, chalk powder – water mixture, etc.
Question 5.
Classify each of the following as a homogeneous or heterogeneous mixture :
soda water, wood, air, soil, vinegar, filtered tea.
Answer:
Soda wafer – Homogeneous
Wood – Heterogeneous
Air – Heterogenous
Soil – Heterogeneous
Vinegar – Homogeneous
Filtered tea – Homogeneous.
Question 6.
How woiild you confirm that a colourless liquid given to you is pure water ?
Answer:
We can confirm that the colourless liquid given to us is pure water by checking its boiling point. Pure water will boil at 373 K at normal atmospheric pressure (1 atm) but water containing some salt will boil oyer a range of temperature above 373 K.
Question 7.
Which of the following materials fall in the category of a ‘‘pure substance” :
(а) Ice
(b) Milk
(c) Iron
(d) Hydrochloric acid
(e) Calcium oxide
(f) Mercury
(g) Brick
(h) Wood
(i) Air
Answer:
Iron, hydrochloric acid, calcium oxide and mercury are pure substances.
![]()
Question 8.
Identify the solutions among the following mixtures:
(a) Soil
(b) Sea water
(c) Air
(d) Coal
(e) Soda water
Answer:
Sea water and soda water are solutions.
Question 9.
Which of the following will show “Tyndall effect”?
(a) Sait solution
(b) Milk
(c) Copper sulphate solution
(d) Starch solution
Answer:
Milk and starch solution will show “Tyndall Effect”.
Question 10.
Classify the following into elements, compounds and mixtures:
(a) Sodium
(b) Soil
(c) Sugar solution
(d) Silver
(e) Calcium carbonate
(f) Tin
(g) Silicon
(h) Coal
(i) Air.
(j) Soap
(k) Methane
(l) Carbon dioxide
(m) Blood
Answer:
(a) Element
(b) Mixture
(c) Mixture
(d) Element
(e) Compound
(f) Element
(g) Element
(h) Element
(i) Mixture
(j) Compound
(k) Compound
(l) Compound
(m) Mixture
![]()
Question 11.
Which of the following are chemical changes ?
(a) Growth of a plant
(b) Rusting of iron
(c) Mixing 9f iron filings and sand
(d) Cooking of food
(e) Digestion of food
(f) Freezing of water
(g) Burning of a candle
Answer:
Rusting of iron, cooking of food, digestion of food and burning of a candle are chemical changes.
Bihar Board Class 9 Science Chapter 2 Is Matter Around Us Pure Textbook Activities
Activity 2.1
- Let us divide the class into groups. A, B, C and D.
- Group A takes a beaker containing 50 mL of water and one spatula full of copper sulphate powder. Group B takes 50 mL of water and two spatula full of copper sulphate powder in a beaker.
- Groups C and D can take different amounts of copper sulphate and potassium permanganate or common salt (sodium chloride) and mix the given components to form a mixture.
- Report the observations on the uniformity in colour and texture.
(i) What type of mixture will be obtained by groups A and B ? Will both the groups obtain mixture of the same colour ? What does this indicate ?
(ii) What type of mixture will be obtained By groups C and D ? Give more examples of this type of mixture ?
Answer:
(i) (a) Groups A and B will obtain homogeneous mixtures i.e., a mixture which has a uniform composition throughout.
(b) Both group A and group B will obtain a copper sulphate solution but the intensity of colour will be different. Group A will obtain light blue and group B will obtain dark blue; colour.
(c) This indicates that a mixture can have a variable composition.
(ii) (a) Group’p C and D will obtain heterogeneous mixture i.e., a mixture containing physically distinct parts and having a (Son-uniform composition.
(b) Other examples of heterogeneous mixture are a mixture of sodiumvchloride and iron filings, salt and sulphur, oil and water etc.
Activity 2.2
- Let us again divide the class into four groups – A, B, C and D.
- Distribute the \following samples to each group :
- Few crystals of cbpper sulphate to group A.
- One spatula full oT copper sulphate to group
- Chalk powder or wheat flour to group C.
- Few drops of milk or ink to group D.
(i) Each group should add the given sample in water and stir properly psing a glass rod, Are the particles in the mixture visible ?
(ii) Direct a beam of light from aiorch throughthe beaker containing the mixture and observe frdjnthe front. Was the path ofthe beam of light visible ?
(iii) Leave the mixtures undisturbeckfor a few minutes (and set up the filtration apparatus in the meantime). Is the mixture stable or do the particles begin to settle after some time ?
(iv) Filter the mixture. Is there ahy residue on the filter paper ?
(v) Discuss tlie results and form an opinion.
Answer:
(i) Groups A, B and D will not be able to see the particles. Group C will be able to see the particles.
(ii) The path of light will be visible by group C and D.
(iii) The mixture is stable for A, B and D and unstable for C.
(iv) In group A, B, D no residue will be left. In group C residue will be left on the filter paper.
(v) Groups A and B have got a solution. Group C has got a suspension. Group D has got a colloidal solution.
![]()
Activity 2.3
- Take approximately 50mL of Water each in two separate beakers.
- Add salt in one beaker and sugar or barium chloride in the second beaker with continuous stirring.
- When ao more solute can be dissolved, heat the contents of the beaker.
- Start adding the solute again.
(i) What is your observation?
(ii) Is the amount of salt and sugar that can be dissolved in water at a given temperature same ?
(iii) What do you infer from the above observation ?
Answer:
(i) We will observe that both salt and sugar keep dissolving while stirring upto a certain level. Beyond a particular level, the solute cannot be dissolved.
(ii) No, the amount of salt and sugar that can be dissolved in water at a given temperature is not the same.
(iii) We infer that different substances in a given solvent have different solubility at the same temperature.
Activity 2.4
- Fill half a beaker with water.
- Put a watch glass on the mouth of the beaker(Fig. 2.3)
- Put few drops of ink on the watch glass.
- Now start heating the beaker. We do not want to heat the ink directly. You will see that evaporation is taking place from the watch glass.
- Continue heating as the evaporation goes on and stop heating when you do not see any further change on the watch glass.
- Observe carefully and record your observations.
(i) What do you think has got evaporated from the watch glass ?
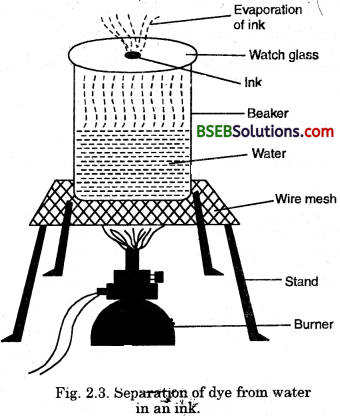
(ii) Is there a residue oh the watch glass ?
(iii) What is your inteiptSsetation ? Is ink a single substance (pure) or is it sjf mixture,?
Answer:
(i) Water has got evaporated from the watch glass.
(ii) Yes, a residue will be left on the watch glass. It will be the dyh of the ink.
(iii) Ink is a mixture of a dye in water.
![]()
Activity 2.5
- Take some full-cream milk in a test tube.
- Centrifuge it by using a centrifuging machine for two minutes. If a centrifuging machine is not available in the school,/you can do this activity at home by using a milk churner, used in the kitchen,
- If you have a milk dairy nearby,visit it and ask (i) how they separate cream from milk and (ii) how they make chease (paneer) from milk.
(i) What do you observe on churning milk ?
(ii) Explain how the separation of cream from milk takes place.
Answer:
(i) On churning milk a layer of cream is formed on top which can be separated.
(ii) This separation has taken place because cream is , lighter than milk. The denser particles of milk are fbrced to the bottom and the lighter cream particles stay at the top when spun rapidly.
Activity 2.6
- Let us try to separate kerosene oil from water using a separating funnel.
- Pour the mixture of kerosene oil and water in a separating funnel Fig. 2.5.
- Let it stand undisturbed for sometime so that separate layers of oil and water are formed.
- Open the stopcock of the separating funnel and pour out the lower layer of water carefully.
- Close the stopcock of the separating funnel – as the oil reaches the stop-cock.
(i) Which liquid forms the lower layer ?
(ii) Which liquid forms the upper layer ?
(iii) What can be inferred from the above observation ?
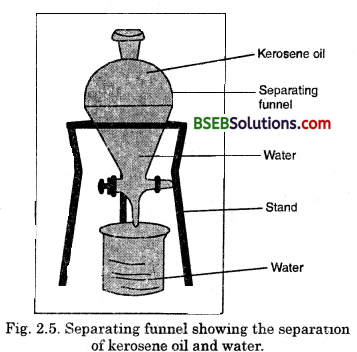
Fig. 2.5, Separating funnel showing the separation of kerosene oil and water.
Answer:
(i) Water forms the lower layer.
(ii) Kerosene oil forms the upper layer.
(iii) We can infer that the heavier liquid (water) forms the lower layer.
![]()
Activity 2.7
- Take a thin strip of filter paper.
- Draw a line on it using a pencil, approximately 3 cm above the lower edge (Fig. 2.7 a).
- Put a small drop of ink (water soluble, that is, from a sketch pen or fountain pen) at the centre of the line. Let it dry.
- Lower the filter paper into a jar/glass/beaker/ test tube containing water so that the drop of ink on the paper is just above the water level, as shown in (Fig. 2.7 and leave it undisturbed).
- Watch carefully, as the water rises up on the filter paper. Record your observations.
(i) What do you observe on the filter paper as the water rises on it ?
(ii) Do yOu obtain different colours on the filter paper strip ?
(iii) What according to you, can be the reason for the rise of the coloured spot on the paper strip ?
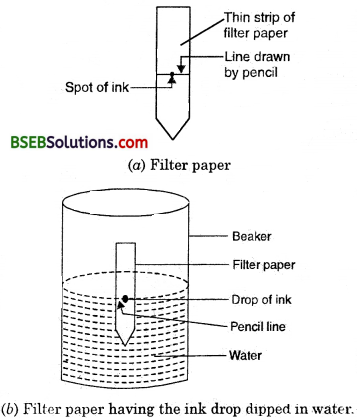
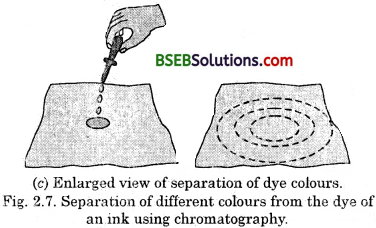
Answer:
(i) As the time passes, the water rises on the filter paper and takes along with it the dye particles. (The ink that we use has water as the solvent and the dye is soluble in it.)
(ii) Yes, we will obtain different colours because dye is, a mixture of two or more colours.
(iii) The coloured component is more soluble in water and hence rises faster in this way the colours get separated.
![]()
Activity 2.8
- Let us try to separate acetone and water from their solution
- Take the mixture in a distillation flask. Fit it with a thermometer.
- Arrange the apparatus as shown in Fig. 2.8.
- Heat the mixture slowly keeping a close watch-at the thermometer.
- The acetone vaporises, condenses in the condenser and can he collected from the condenser outlet.
- Water is left behind in the distillation flask.
(i) Wliat do you observe as you start heating the’ mixture ?
(ii) At what temperature does the thermometer reading become constant for some time ?
(iii) What is the boiling point of acetone ?
(iv) Why do the two components separate ?
(v) What is this process called ?
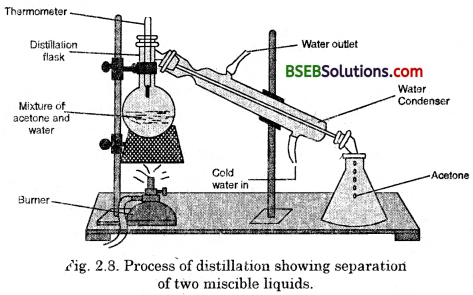
Fig, 2.8. Process of distillation showing separation of two miscible liquids.
Answer:
(i) Some vapours will be evolved (acetone fumes).
(ii) The thermometer reading will become constant for sometime at the boiling point of the mixture. Acetone mixed with water will boil closer to 88-90°C. Depending on the ratio of aicohol and water, the boiling point can be anywhere between 80°C and 100°C.
(iii) The boiling point of absolute acetone is 78.3°C.
(iv) The two components separate because they have sufficient difference in their boiling points.
(v) This process is called simple distillation.
![]()
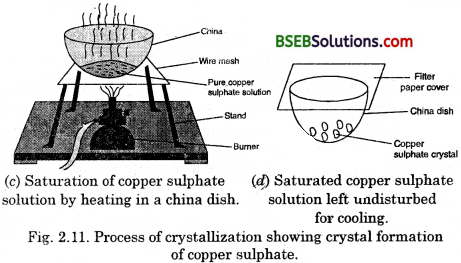
Activity 2.9
- Take some (approximately 5 g) impure sample of copper sulphate in a china dish.
- Dissolve it in minimum amount of water.
- Filter the impurities out.
- Evaporate water from the copper sulphate solution so as to get a saturated solution.
- Cover the solution with a filter paper and leave it undisturbed at room temperature to cool slowly for a day.
- You will obtain the crystals of copper sulphate in the china dish.
- This process is called crystallisation.
(i) What do you observe in the china dish ?
(ii) Do the crystals look alike ? What is this process called ?
(iii) How will you separate the crystals from the liquid in the china dish ?
(iv) What is the need to heat the solution ?
(v) Besides crystallization, which other methods have we used in this activity ?
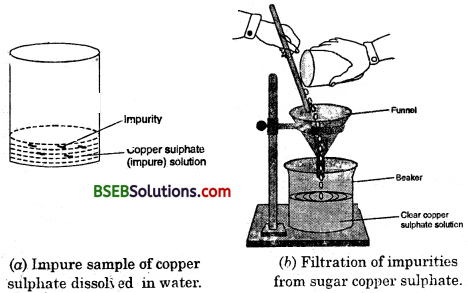
Answer:
(i) We will obtain crystals of copper sulphate in the china dish.
(ii) The crystals don’t look alike. This processes called crystallisation.
(iii) The crystals from the liquid in the china dish can be separated by the process of decantation.
(iv) The solution is heated to saturate it.
(v) Besides crystallization we have used filtration and decantation.
Activity 2.10
- Divide the class into two groups. Give 50 grams of iron filings and 3 g of sulphur powder in a china dish to both the groups.
Group I
- Mix and crush iron filings and sulphur powder.
Group II
- Mix and crush iron filings and sulphur powder. Heat this mixture strongly till red hot. Remove from flame and let the mixture cool.
Groups I and II
- Check for magnetism in the material obtained. Bring a magnet near the material and check if the material is attracted towards the magnet.
- Compare the texture and colour of the material obtained by the groups.
- Add carbon disulphide to one part of the material obtained. Stir well and filter.
- Add dilute sulphuric acid or dilute hydrochloric acid to the other part of the material obtained. (Note: teacher supervision is necessary for this activity).
Perform all the above steps with both the elements (iron and sulphur) separately.
(i) Did the material obtained by both the groups look the same ?
(ii) Are we able to separate the components of the material ?
(iii) On adding dilute sulphuric acid, did both the groups obtain a gas ? Did the gas in both the cases smell same or different ?
(iv) Material obtained by which group has magnetic properties ?
(v) What type of change lias taken place in the two cases ?
(vi) What can we infer from the above activity ?
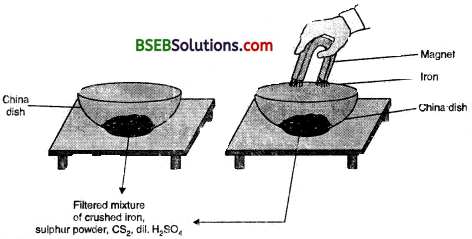
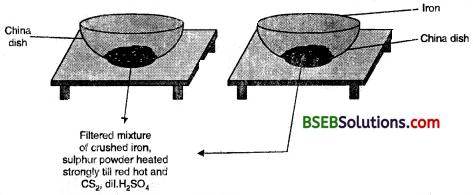
Answer:
(i) No, the material obtained by both the groups did not look the same.
(ii) In group I, the components can be separated by physical methods and in group II, the components can be separated by chemical methods.
(iii) Yes, both the groups obtained a gas on adding dilute sulphuric acid. The gas obtained by group I is hydrogen, which is colourless, odourless and combustible. The gas obtained by group II is hydrogen sulphide (H2S), which is colourless with a smell of rotten eggs.
(iv) Group I will show magnetic properties.
(v) In case of group I, a physical change has taken place whereas in case of group II, a chemical change has taken place.
(vi) We can infer that group I has obtained a mixture and group II has obtained a compound. The properties of the mixture are the same as that of its constituents whereas the compound obtained by group II has totally different properties compared to the combining elements.
![]()
Bihar Board Class 9 Science Chapter 2 Is Matter Around Us Pure Additional Important Questions and Answers
Very Short Answer Type Questions
Question 1.
What is a substance ?
Answer:
A substance is a kind of matter that cannot be separated into other kinds of matter by any physical process. ;
Question 2.
Are the naturally occurring materials on earth substances ? ,
Answer:
No, most of the naturally occurring materials on earth are mixtures.
Question 3.
What does the word pure written 6n;a packet of.milk, ghee (butter), fruit juice etc. mean for a common person ?
Answer:
The word pure written on food packets means that there is no adulteration in the food item.
Question 4.
Write three constituents of milk.
Answer:
Milk is a mixture of water, fat and protein.
Question 5.
What does a scientist mean when he says something is pure ?
Answer:
When a scientist says something is pure he means that it is a single substance.
Short Answer Type Questions
Question 1.
What is a mixture ?
Answer:
When two or more substances (elements or compounds or both) are combined physically in any proportion such that each constituent retains its chemical property, then the resulting substance is called a mixture.
Question 2.
Give two examples of mixture.
Answer:
Air, sugar solution.
Question 3.
What are the two types of mixtures ?
Answer:
Homogeneous mixture and heterogeneous mixture.
Question 4.
What is a homogeneous mixture ?
Answer:
A mixture having uniform composition throughout its mass is known as homogeneous mixture.
Long Answer Type Question
Question 1.
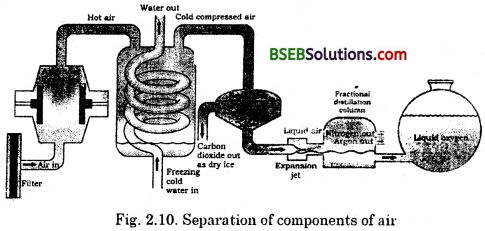
How can we obtain oxygen gas from air ?
Answer:
If we want oxygen gas from air (Fig. 2.10), we have to separate all the other gases present in the air. The air is compressed by increasing the pressure and is then cooled by decreasing the temperature to get liquid air. This liquid air is allowed to warm-up slowly in a fractional distillation column, where gases get separated at different heights depending upon their boiling points.